Neuroimaging Biomarkers in Alzheimer Disease and Related Dementias: From Structural MRI to Amyloid and Tau PET
This practical guide explains how multimodal neuroimaging refines the diagnosis of Alzheimer disease, frontemporal dementia, dementia with Lewy bodies, and vascular dementia.
Neurocognitive disorders are a common cause of morbidity and mortality worldwide, particularly in older populations. Starting with the introduction of CT in the 1970s, neuroimaging has been increasingly valuable in the assessment of individuals with cognitive symptoms. Commercially available neuroimaging modalities are sensitive to neurodegeneration (MRI and 18F-fluorodeoxyglucose [FDG] positron emission tomography [PET]); Β-amyloid deposition (amyloid PET); and amyloid-related imaging abnormalities (ARIA), the characteristic complications of amyloid-targeting therapies. Tau PET, which has been approved by the Food and Drug Administration (FDA) but is not yet widely available, provides a biologic marker of Alzheimer disease (AD) progression.
Structural Imaging (CT and MRI)
In individuals presenting with dementia, structural imaging modalities such as noncontrast CT or MRI are usually performed as part of the initial evaluation. Although CT lacks the resolution and tissue-specific imaging sensitivities of MRI, clinically significant mass lesions that may require surgical intervention, such as subdural hematomas, neoplasms, and hydrocephalus, can be readily visualized with routine CT. Furthermore, CT, which can be completed in seconds, is particularly valuable in individuals with advancing dementia who may not tolerate the relatively prolonged immobilization required for MRI.
With its superior resolution and standard multiplanar capability, MRI is the preferred initial imaging modality in the evaluation of individuals with suspected cognitive disorders. In contrast to traditional CT protocols, standard brain MRI should include sagittal, coronal, and axial sequences. Most MRI protocols include sequences that are sensitive to edema (fluid-attenuated inversion recovery [FLAIR] and T2-weighted sequences), acute ischemia or cortical vacuolar degeneration (diffusion-weighted sequences), blood products (gradient echo or susceptibility-weighted sequences), and other pathologic processes. If neoplasm, intracranial infection, or demyelination are suspected, contrast-enhanced MRI sequences should be strongly considered. MRI is contraindicated when individuals have ferromagnetic materials in their body, such as implanted defibrillators, cochlear implants, or bullet fragments or other shrapnel.
Commercially available software can automatically calculate volumes of hippocampi and other structures, but these packages have not been widely implemented outside major academic institutions. MRI-based visual rating scales using routine clinical sequences, such as entorhinal cortex atrophy (ERICA) and medial temporal atrophy (MTA) scales (Figure 1), are sometimes used as auxiliary biomarkers of neurodegeneration in AD.1 By assessing several medial temporal lobe structures, the MTA scale has been demonstrated to provide good discrimination between individuals with AD and healthy controls in a memory clinic.2 The ERICA score rates the volumes of the entorhinal and parahippocampal gyrus. Another rating tool, the Fazekas scale, rates the severity of white matter disease, which is commonly seen in individuals with vascular cognitive impairment or other clinical scenarios.
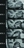
Figure 1. Medial temporal atrophy (MTA) rating scale. Cropped T1-weighted coronal images of the right medial temporal lobes demonstrate progressive hippocampal atrophy and corresponding enlargement of the adjacent portion of the right lateral ventricle. From Flodin FF, Haller S, Poom L, Fällmar D. Congruency between publicly available pictorial displays of medial temporal lobe atrophy. Eur Radiol. 2025;35(10):5944-5953.1 Used with permission (http://creativecommons.org/licenses/by/4.0).
MRI studies often demonstrate clinically relevant patterns of atrophy, which can be readily evident even through casual visual inspection. Figure 2 demonstrates atrophy which is maximal in the anterior left temporal lobe, commonly described as knife-like gyri, in an individual with the semantic dementia subtype of frontotemporal dementia (FTD). Other characteristic patterns of atrophy that may be evident on MRI include midbrain atrophy (the hummingbird sign) in individuals with progressive supranuclear palsy, atrophy of the caudate nuclei in individuals with Huntington disease, and asymmetric supratentorial hemispheric atrophy in individuals with corticobasal degeneration.

Figure 2. Axial fluid-attenuated inversion recovery MRI demonstrates prominent atrophy of the left anterior temporal lobe (arrow) in an individual with progressive aphasia (age mid-60s) (A). The coronal T2-weighted image demonstrates knife-like gyri (arrowheads) in the atrophic left temporal lobe (B).
Molecular Imaging
Molecular imaging, mostly using PET, has revolutionized the diagnosis and treatment of AD by providing a positive diagnosis, rather than one of exclusion, and guiding the development and clinical trials of the first medications shown to improve the evolution of the disease.3,4 Clinically useful PET modalities include metabolic, amyloid, and tau imaging.
Metabolic Imaging
Regional brain metabolism, measured with 18F-FDG PET, is indicated to differentiate AD from FTD, which often also presents with memory impairment.5 Unlike AD, which tends to affect posterior brain regions, the anterior temporal and frontal lobes are usually hypometabolic in FTD.6 However, individuals with FTD, particularly those with progranulin or certain MAPT gene mutations. may also demonstrate decreased parietal metabolism.7 The most typical metabolic pattern found in early AD is decreased metabolism bilaterally in the parieto-temporal association cortex and posterior cingulate gyrus8 (Figure 3). As AD progresses, some areas of the frontal association cortex become hypometabolic, whereas the paracentral cortex (primary motor-sensory areas) has normal metabolism (Figure 4).

Figure 3. 18F-fluorodeoxyglucose positron emission tomography findings in diseases leading to dementia—including Alzheimer disease; frontotemporal dementia (FTD), behavioral variant; FTD, agrammatic primary progressive aphasia (PPA) variant; and semantic dementia—projected on MRI scans from the same individual.

Figure 4. Metabolism, amyloid, and tau imaging in an individual with logopenic aphasia associated with Alzheimer disease, age late 50s. The primary sensory-motor areas (asterisks), as well as the primary visual (striatal cortex) and auditory (Heschl gyrus) regions (arrowheads), have normal metabolism and no tau deposition. By contrast, areas with high tau deposition (eg, the inferior parietal lobule [arrows]) tend to have decreased metabolism. In some areas, high amyloid deposition corresponds to low metabolism and increased tau (eg, the precuneus). However, there are areas with high amyloid load and normal metabolism, such as the medial occipital region. Uptake in the region of the substantia nigra does not correspond with tau deposition. From Pascual B, Masdeu JC. Tau, amyloid, and hypometabolism in the logopenic variant of primary progressive aphasia. Neurology. 2016;86(5):487-488. Used with permission.
The metabolic pattern found in AD can also be found in those with diffuse Lewy body disease (dementia with Lewy bodies [DLB]), in part because the 2 brain pathologies often coexist.9,10 In contrast to those living with AD, individuals with DLB often have retained activity in the posterior cingulate gyrus, giving rise to the posterior-cingulate island sign on FDG PET11 (Figure 5). DLB pathology can also be distinguished from AD using dopamine transporter single-photon emission CT imaging studies, which commonly have abnormal results in DLB. An algorithm using 18F-FDG PET has been shown to have a strong predictive value in differentiating brain neurodegenerative disorders,12 and has the potential to support diagnoses in combination with reasoning from clinical findings.

Figure 5. The island sign in dementia with Lewy bodies (DLB). On MRI templates of the medial aspect of the brain, areas of decreased metabolism (18F-fluorodeoxyglucose positron emission tomography) can be seen in Alzheimer disease (AD) (A) and decreased perfusion (H215O positron emission tomography) can be seen in DLB (B). Metabolism and perfusion are coupled in AD and DLB. Note involvement of the posterior cingulate gyrus in AD but sparing of this region (arrow) in DLB. From Goker-Alpan O, Masdeu JC, Kohn PD, et al. The neurobiology of glucocerebrosidase-associated parkinsonism: a positron emission tomography study of dopamine synthesis and regional cerebral blood flow. Brain. 2012;135(Pt 8):2440-2448. Used with permission.
Amyloid Imaging
Amyloid deposition is the strongest and earliest neuroimaging predictor of future cognitive impairment and progression from mild cognitive impairment (MCI) to AD, conferring a 3- to 7-fold increased risk compared with individuals without amyloid deposition.13-15 Amyloid imaging is also a powerful tool to differentiate the dementias characterized by amyloid deposition, such as AD and diffuse DLB (which is often associated with AD9), and non–amyloid-related disorders, such as FTD.16 However, some older individuals with FTD may have coexistent amyloid brain deposition simply due to advancing age.17,18
Good concordance with histologically measured amyloid load has been shown for three 18F amyloid PET tracers that are approved by the FDA for use in the clinical setting: 18F-florbetapir,19 18F-flutemetamol,20 and 18F-florbetaben.21 A fourth amyloid PET tracer, 18F-flutafuranol, also known as 18F-NAV4694, is used in research. 18F-flutafuranol has much less white matter binding than other 18F tracers, thus providing cleaner images, which is particularly helpful in individuals with incipient amyloid deposition.22 Demonstrated amyloid positivity by either lumbar puncture or amyloid PET is required under current FDA label approvals to prescribe antiamyloid medications. Most US clinicians recommend amyloid PET instead of a lumbar puncture.23
Brain amyloid deposition begins in the presymptomatic stages of AD,24 increases during the MCI stage, and remains relatively stable as the disease progresses.25 Thus, amyloid deposition serves as a marker of the presymptomatic stages of the disease and correlates with the degree of cognitive impairment in the preclinical stages and in MCI, but not in AD25,26; however, atrophy on MRI and synaptic dysfunction on 18F-FDG PET continue to increase and spread as clinical AD worsens and cognition deteriorates.25
In early AD, amyloid deposition is highest in the default network and, thus, in the fronto-parieto-temporal association cortex, including the precuneus, but spares the paracentral regions and primary visual and auditory sensory cortex (Figure 4). The caudate nucleus is often affected as well.
Tau Imaging
In the healthy brain, the tau protein stabilizes neurotubules and is therefore essential for normal neural function. However, in AD and other neurodegenerative disorders, tau becomes abnormally hyperphosphorylated, dysfunctional, and misfolded, constituting the intraneuronal tangles observed neuropathologically in AD and other tauopathies. PET tracers are available that bind strongly to the abnormally folded tau (also referred to as hyperphosphorylated tau, or simply tau). These tracers do not bind to the healthy, native form of tau. The only PET tracer approved by the FDA for tau imaging in AD is 18F-flortaucipir, which has been shown in postmortem studies to bind to tau tangles composed of 3- and 4-repeat tau aggregates.27 18F-flortaucipir binds much less, or not at all, to 3- or 4-repeat tau aggregates found in most varieties of tau-related FTD.28,29 Binding is most likely determined by the configuration of tau aggregates, which differs in various tauopathies.30 For instance, 18F-flortaucipir binds to tau aggregates in individuals harboring a p.R406W mutation in the MAPT gene which encodes tau.31 This variant results in 3- and 4-repeat tau aggregates, similar to those seen in AD.32
18F-flortaucipir shows highly specific uptake in areas known neuropathologically to contain a large amount of tau in AD32-34 (Figure 4). It exhibits minimal white matter binding; however, uptake is observed in the substantia nigra and the choroid layer of the eye, likely reflecting binding to melanin,28,35 and in the choroid plexus, possibly due to binding to calcifications or tau in this structure.29,36 In older individuals, even those without cognitive impairment, nonspecific binding may occur in the lenticular nucleus, red nucleus, subthalamic nucleus, and anterior cerebellar lobule.
Because antiamyloid therapies modify symptom progression only in individuals with a small amount of brain tau,37 tau imaging shows potential to select those likely to benefit from these therapies. However, the amount or degree of tau spreading that would contraindicate prescription of antiamyloid monoclonal therapy has not been determined, and could differ among available monoclonal antibodies.
Tau accumulation measured with tau PET tracers correlates better with the degree of cognitive impairment than amyloid accumulation,38 which is in agreement with neuropathologic studies.39 Furthermore, there is an inverse correlation between tau accumulation and brain metabolism; regions high in tau have uniformly depressed metabolism34 (Figure 4). This correlation does not occur with amyloid accumulation (Figure 4).
Multimodal Imaging
Whereas noncontrast MRI is generally the initial imaging study used to assess individuals with neurocognitive disorders, PET and other imaging modalities also have clinical utility. The American College of Radiology maintains imaging guidelines41 that provide recommendations about the appropriateness of various imaging modalities in the evaluation of common and uncommon clinical phenotypes of cognitive dysfunction (Table 1).

ARIA Monitoring
The recent FDA approval of antiamyloid monoclonal antibodies (ie, amyloid targeting therapy)42,43 added new options to treat early-stage AD. Leqembi (lecanemab; Eisai, Nutley, NJ) targets soluble amyloid protofibrils as well as insoluble amyloid plaques. Kisunla (donanemab; Eli Lilly, Indianapolis, IN) is directed against amyloid plaques. For the neuroimager, the most notable potential adverse effect associated with these agents is ARIA.44 Although usually asymptomatic, severe or (very rarely) fatal cases have occurred.
Amyloid can be deposited not only in the parenchyma but also in the wall of cerebral vessels (ie, amyloid angiopathy). Antiamyloid antibodies also affect these vascular amyloid deposits, which may further compromise the integrity of the already weakened vessel wall. This can lead to extravasation of fluid (edema) or red blood cells (hemorrhage); thus, subtypes of ARIA include ARIA-E (with edema) and ARIA-H (hemorrhagic).44,45 These may occur in isolation, but more often appear in combination.46 They can occur at any time during therapy, but are more likely during the early course (up to 6 to 9 months), after which the incidence decreases.47 During this more vulnerable time, individuals need to be under regular MRI surveillance, even if asymptomatic, to monitor for potential ARIA.
On the MRI scan, ARIA-E appears as a single area or multiple areas of T2/FLAIR hyperintense signal change of variable extent (Figure 6). The most common locations are the occipital lobes, followed by frontal, parietal, and temporal lobes, but the cerebellum may also be involved.44,46 Although typically confined to the white matter, the edema can also affect the gray matter, appearing as gyral swelling. At times, leakage of proteinaceous fluid into the leptomeningeal/subpial space causes sulcal FLAIR hyperintensity (Figure 7), which resembles focal subarachnoid hemorrhage.44,46 In these cases, however, susceptibility-weighted imaging does not reveal the presence of blood products.44 ARIA-E does not cause restricted diffusion, which helps differentiate it from acute ischemia.46
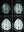
Figure 6. Amyloid-related imaging abnormalities, both with edema and microhemorrhages, after the 5th infusion of antiamyloid therapy. On the initial scan, there is radiographically mild vasogenic edema in the right frontal lobe (A). On the follow-up scan 3 months later, the edema has resolved (B). Intermixed with the edema, on the initial scan, radiographically severe microhemorrhages are present: their number, including adjacent slices, exceeds 10 (C). On the 3-month follow-up scan, the microhemorrhages are stable (D).

Figure 7. Sulcal or effusion amyloid-related imaging abnormalities with edema. Increased signal is observed in the left intraparietal sulcus (arrows) after 4 infusions. The normal intraparietal sulcus is shown on the right.
ARIA-H can present with various imaging manifestations. Most commonly, microhemorrhages occur, often colocalizing with ARIA-E (Figure 6). Superficial siderosis is a less common finding (Figure 8). Rarely, larger intracerebral hemorrhages (>1 cm) have been reported.48

Figure 8. Amyloid-related imaging abnormalities, hemorrhagic type, as a result of antiamyloid infusion therapy, manifesting as superficial siderosis involving adjacent sulci of the left frontal lobe (double arrows).
The role of the neuroimager is to recognize the presence and severity of ARIA, as summarized in Table 2.48 If ARIA is detected, follow-up imaging is usually needed to ensure resolution of ARIA-E, which usually occurs in 1–4 months,47 and to monitor for the progression of ARIA-H. The severity of the clinical and radiographic findings dictates whether treatment may continue, is put on hold, or must be permanently discontinued.

At a minimum, the ARIA imaging protocol should include axial FLAIR sequences and additional sequences for hemorrhage detection. Susceptibility-weighted imaging is preferred over gradient echo, given its greater sensitivity for blood detection. Diffusion-weighted imaging is also recommended, to differentiate edema from acute ischemia in questionable cases. To ensure accurate comparisons, serial MRI monitoring should be carried out on the same scanner (or at least a scanner with the same field strength) with the same protocol. Scanning using a 3T magnet is preferred.
Besides traditional interpretation of the imaging studies, tools powered by artificial intelligence may enhance the sensitivity and precision of ARIA detection and monitoring49 and can be especially helpful in identifying subtle edematous changes.
The presence of ARIA-E may be linked to adverse clinical outcomes. However, in a recent study using amyloid PET imaging before and after moderate to severe ARIA-E, significantly greater PET signal reduction was found in the affected regions after edema resolution,50 suggesting that the presence of ARIA-E corresponds to robust clearance of Β-amyloid.
Conclusions
Brain MRI remains the most appropriate initial imaging modality in the evaluation of most individuals with neurocognitive disorders. Characteristic findings are commonly observed across different neurocognitive disorders, confirming the role of structural neuroimaging in individuals with diverse phenotypes. Nonetheless, multimodal imaging has become commonplace in large part due to the progressive adoption of amyloid-targeting therapies for individuals with AD. Whereas visual analysis of MRI sequences can identify changes commonly seen in AD and other disorders, nuclear medicine studies are necessary to confirm the underlying metabolic signature of disease, or, in the case of AD, the abnormal accumulation of Β-amyloid or tau. Serial MRI testing is mandated by the FDA to identify symptomatic and asymptomatic ARIA-E and ARIA-H in individuals receiving amyloid-targeting therapies.
Ready to Claim Your Credits?
You have attempts to pass this post-test. Take your time and review carefully before submitting.
Good luck!
Recommended
- Imaging & Testing
MRI Clues to Intracranial Infection: Differentiating Bacterial, Viral, Fungal, and Parasitic CNS Disease
Márk Kozák, MD; Máté Sik, MDMárk Kozák, MD; Máté Sik, MD - Imaging & Testing
Practical MRI in Neurology: Use Cases, Technologies, and Opportunities for Outpatient Clinics
Joseph Fritz, PhD; Edmond A. Knopp, MD, DABR, FASFNRJoseph Fritz, PhD; Edmond A. Knopp, MD, DABR, FASFNR - Imaging & Testing
Neuroimaging After Brain Radiotherapy and Radiosurgery
Venkatesh Shankar Madhugiri, MBBS, MCh, FACS; Neil D. Almeida, MDVenkatesh Shankar Madhugiri, MBBS, MCh, FACS; Neil D. Almeida, MD



