MRI Clues to Intracranial Infection: Differentiating Bacterial, Viral, Fungal, and Parasitic CNS Disease
A practice-focused neuroimaging approach highlights how diffusion, contrast enhancement, and pattern recognition on MRI can directly influence prognosis and treatment decisions in CNS infections.
Infections involving the central nervous system (CNS) represent some of the most challenging conditions in neurology, given their rapid progression, high morbidity, and potential for irreversible injury. Neuroimaging has become indispensable in the diagnosis, management, and follow-up of neuroinfections, providing critical information that supplements the clinical and laboratory findings.1
CT and MRI are the primary cross-sectional imaging modalities used to assess the extent and severity of CNS infections. CT is preferred for unstable individuals, for quickly ruling out hemorrhage or hydrocephalus, and for surgical planning in cases of bone involvement; however, MRI is favored for nearly all diagnostic evaluations, especially for parenchymal, pachymeningeal, or leptomeningeal disease.1,2 Contrast administration is recommended unless contraindicated (eg, severe renal impairment, previous severe contrast reaction), because contrast-enhanced MRI is particularly helpful for delineating infectious from noninfectious processes, with characteristic findings such as leptomeningeal or rim enhancement aiding in differentiating diffuse inflammation from well-formed abscesses.3 Standard and adjunct MRI sequences, including T1- and T2-weighted imaging, fluid-attenuated inversion recovery (FLAIR), susceptibility-weighted imaging (SWI), diffusion-weighted imaging (DWI), apparent diffusion coefficient (ADC) mapping, and magnetic resonance spectroscopy, enhance diagnostic accuracy and can identify complications such as infarction or ventriculitis.4
Neuroimaging also plays a prognostic role. Abnormal MRI findings, such as cerebral ischemia, extensive edema, or hydrocephalus, have been independently associated with unfavorable outcomes and increased mortality risk, particularly in bacterial meningitis and tuberculous CNS infections.5 Early recognition of these changes can expedite timely surgical or critical care interventions. Despite technologic advances, interpreting imaging findings remains complex, especially in immunocompromised individuals, where atypical pathogens or overlapping autoimmune and neoplastic mimics are common.5
This review summarizes the characteristic imaging appearances of common CNS infections along with their complications and mimics. Emphasis is placed on practical diagnostic strategies relevant to everyday neurologic practice. Our goal is to summarize the most typical radiologic findings of the most common CNS infections, highlighting imaging clues that guide clinical decision-making.
Imaging Findings in Neuroinfections
Bacterial Infections
Pyogenic Meningitis. A large epidemiologic study using US registries estimates the incidence of bacterial meningitis at 0.7 to 1.3 per 100,000 individuals.6 In adults, Streptococcus pneumoniae and Neisseria meningitidis are the predominant pathogens implicated. The most common route of infection is hematogenous spread of bacteria to the meninges, but direct extension from extracranial infection—most commonly from sinusitis or otitis media—is also possible.7
When neuroimaging is indicated before lumbar puncture, a noncontrast head CT is usually obtained first. Although CT has limited diagnostic value for meningitis, it is useful for excluding space-occupying lesions with significant mass effect, obstructive hydrocephalus, or acute intracranial hemorrhage. Occasionally, CT may demonstrate sulcal narrowing or effacement as an early sign of cerebral edema; however, this finding is neither sensitive nor specific to meningitis.8
On MRI, the hallmark feature of meningitis is leptomeningeal enhancement, reflecting increased blood–brain barrier permeability due to inflammation (Figure 1). Meningeal enhancement can present as linear or nodular signal changes along the pial surface. Contrast-enhanced 3D FLAIR demonstrates greater sensitivity for detecting meningeal involvement compared with postcontrast T1-weighted imaging.9 Subarachnoid purulent infection may demonstrate restricted diffusion. Altered cerebrospinal fluid signal can also be detected on FLAIR. In more severe cases, inflammation may extend into the ventricular system, resulting in ventriculitis, characterized by ependymal contrast enhancement and layering intraventricular debris with restricted diffusion on DWI.10

Figure 1. Imaging hallmarks of brain abscess and meningitis. An axial noncontrast T1-weighted image demonstrates a focal parenchymal lesion consistent with abscess (arrowhead) (A). An axial postcontrast T1-weighted image shows smooth ring enhancement of the abscess (arrowhead) with adjacent leptomeningeal enhancement (arrow), consistent with associated meningitis (B). An axial T2-weighted image demonstrates hyperintensity within the abscess (arrowhead) and surrounding vasogenic edema (C). Diffusion-weighted imaging (DWI) shows marked diffusion restriction within the abscess (high signal; boxed area) (D). The apparent diffusion coefficient (ADC) map confirms true restriction (low signal; boxed area) (E). A coronal fluid-attenuated inversion recovery (FLAIR) image demonstrates hyperintensity of the lesion and perifocal edema (F).
Cerebritis and Brain Abscess. On CT, early parenchymal brain infection (ie, cerebritis) appears as patchy unifocal or multifocal hypodensities. Contrast enhancement may be present, particularly on the pial border of the infection, suggesting concomitant meningitis.10 On MRI, the involved parenchyma is typically hyperintense on T2-weighted and FLAIR images. In the early phase, diffusion findings are dominated by vasogenic edema, typically manifesting as low DWI signal and high ADC values. As necrosis develops and a central cavity forms, purulent material shows true diffusion restriction with high DWI signal and low ADC values. A typical mature brain abscess has a T2-hypointense capsule and shows a dual rim sign on SWI (ie, an outer hypointense rim and an inner hyperintense rim). The capsule typically demonstrates thin, smooth contrast enhancement and represents a classic ring-enhancing lesion10,11 (Figure 2).

Figure 2. Solitary pyogenic brain abscess with restricted diffusion. An individual with emphysema and alcohol abuse, age mid-60 years, presented with a 1-week history of severe headaches. Complete resolution was achieved after surgical drainage and antibiotic therapy. Diffusion-weighted imaging demonstrates restricted diffusion within the abscess, consistent with viscous purulent content (A and B). A sagittal fluid-attenuated inversion recovery image shows an ovoid abscess with associated maxillary inflammatory disease (C). An axial postcontrast T1-weighted image demonstrates smooth ring enhancement of the abscess (D).
Subdural and Epidural Empyema. Epidural empyema—a pyogenic collection between the skull and dura mater—most commonly occurs due to direct spread of infection from the paranasal sinuses, middle ear, or mastoid.12 On imaging, these collections are typically biconvex (ie, lentiform), which is an important differential feature. Subdural collections appear crescentic on CT and MRI, and the most useful sign favoring infection is diffusion restriction. However, sedimented or aggregated portions of a noninfectious hemorrhage may also show restricted diffusion13 (Figure 3).

Figure 3. Subdural empyema with concomitant frontal lobe abscess. An individual with HIV infection and a history of intravenous drug use, age early 40 years, previously treated for pneumococcal pneumonia and lung abscess, presented with headache, confusion, and new-onset seizure. Diffusion-weighted imaging demonstrates restricted diffusion consistent with subdural empyema and a left frontal lobe abscess (A). A T2-weighted image confirms a rounded purulent abscess with a hypointense rim (B). Postcontrast T1-weighted imaging shows rim enhancement of the abscess (C).
Viral Infections
Herpes Simplex Encephalitis. In the United States, the incidence of viral encephalitis is estimated to be between 2.5 and 7.5 cases per 100,000 individuals. Among viral infections, herpes simplex encephalitis (HSE) is the most common cause of encephalitis with an associated mortality rate of 70% in untreated individuals.14 HSE may result from viral reactivation or primary infection and is most commonly caused by herpes simplex virus (HSV) type 1 infection (~90%), with HSV type 2 infection accounting for the remaining cases. The lack of typical symptomatology underscores the importance of neuroimaging in the diagnosis of this type of infection. Infectious lesions associated with HSE show a characteristic predilection for the limbic system, particularly the medial temporal lobes, insula, and cingulate gyrus. On MRI, hyperintense signals are visible on T2-weighted and FLAIR sequences, indicating edema related to inflammation (Figure 4). The edema may be asymmetrical but can later extend to both temporal lobes. On T1-weighted images, affected areas appear hypointense due to vasogenic edema. After contrast administration, enhancement typically becomes more conspicuous as the infection progresses, whereas DWI commonly demonstrates restricted diffusion. Hemorrhagic changes are also frequent and may cause blooming artifacts on SWI.15

Figure 4. Neuroimaging findings associated with herpes simplex encephalitis. MRI demonstrates characteristic involvement of the left temporal lobe. A noncontrast T1-weighted image shows left temporomedial cortical swelling and widening (arrow) (A). A postcontrast T1-weighted image demonstrates gyral cortical enhancement (arrow) (B). A T2-weighted image shows cortical and subcortical hyperintensity with gyral swelling (C). Diffusion-weighted imaging (DWI) demonstrates cortical diffusion restriction (arrow) (D). An apparent diffusion coefficient (ADC) map shows corresponding low values (arrow) (E). A fluid-attenuated inversion recovery (FLAIR) image demonstrates hyperintensity with sulcal effacement in the left temporal lobe (F).
Varicella-Zoster Virus Encephalitis. Varicella-zoster virus (VZV) is the causative agent of both primary varicella (ie, chickenpox) and herpes zoster. CNS involvement occurs in a small proportion of infections, estimated at ~0.25%.16 In these rare cases, reactivation of the virus can lead to neurologic complications, such as VZV vasculopathy, encephalitis, or Ramsay Hunt syndrome. VZV-induced vasculopathy involves inflammation of the cerebral arteries, which can result in ischemic or hemorrhagic stroke.
The radiologic features of VZV encephalitis are variable, and their interpretation may be challenging, with diffuse or multifocal involvement of brain regions occurring in a manner similar to HSV. A key distinguishing feature is frequent cerebellar involvement.15,16 General imaging features of inflammation include increased T2-weighted and FLAIR signal intensity, low T1 signal, and subtle contrast enhancement, with variable DWI findings.16 Given the frequent involvement of cerebral arteries, magnetic resonance angiography should be considered as part of the imaging protocol, because it can detect vasculitic changes, vessel narrowing, or occlusion, which have important diagnostic and prognostic implications.16
Cytomegalovirus Encephalitis. Cytomegalovirus (CMV) can cause severe infections, particularly in immunocompromised individuals, such as those with human immunodeficiency virus infection or acquired immunodeficiency syndrome (AIDS) or those receiving immunosuppressants due to organ transplantation or another diagnosis.17 CMV can affect various systems, including the respiratory system, the digestive system, and the CNS. In individuals with compromised immune systems, CMV typically leads to ventriculoencephalitis.15 MRI scans demonstrate periventricular hyperintense T2/FLAIR signals, which are indicative of edema. Thickening of the ventricular walls and contrast enhancement may be observed.16
SARS-CoV-2 Encephalitis. SARS-CoV-2 infection has been associated with a range of neurologic complications, including encephalitis, microvascular injury, and demyelination. Imaging findings are largely nonspecific and overlap with those observed in other viral CNS infections, such as HSV or West Nile virus encephalitis, and typically include increased T2-weighted and FLAIR signal intensity, low T1 signal, and variable diffusion-weighted imaging abnormalities. In the context of SARS-CoV-2 infection, however, these radiologic changes may also reflect secondary mechanisms, including hypoxic–ischemic injury, endothelial dysfunction, microvascular thrombosis, or immune-mediated inflammation, rather than direct viral encephalitis.18
CNS Tuberculosis. CNS tuberculosis, a major problem in endemic regions, can be challenging to diagnose in settings with lower tuberculosis prevalence. Clinical manifestations are frequently related to communicating hydrocephalus, cranial nerve involvement, or ischemic complications secondary to tuberculous arteritis.19 Parenchymal manifestations include cerebritis and granulomas (ie, tuberculomas).
On imaging, CNS tuberculosis most commonly demonstrates basilar cistern–predominant leptomeningeal enhancement with gelatinous exudates. Tuberculomas typically appear as T2-hypointense lesions with ring enhancement; diffusion restriction is less frequent than in pyogenic abscesses. Magnetic resonance spectroscopy often shows lipid and lactate peaks in tuberculomas, whereas normal brain tissue demonstrates a high N-acetylaspartate peak with low lipid and lactate signals. Immunocompromised individuals may develop tuberculous abscesses, which resemble other bacterial abscesses and usually demonstrate diffusion restriction19 (Figure 5).
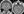
Figure 5. Central nervous system tuberculosis with miliary abscesses and basilar meningitis. Postcontrast T1-weighted images of an individual with acquired immunodeficiency syndrome, hepatitis, and central nervous system tuberculosis. The coronal image demonstrates multiple small miliary parenchymal abscesses consistent with indolent tuberculous infection (arrow) (A). The axial image shows basilar leptomeningeal enhancement compatible with tuberculous meningitis (arrows) (B).
Fungal Infections
Fungal infections of the CNS are most commonly observed in individuals with compromised immune systems, including people with AIDS, diabetes, or immunosuppression due to organ transplantation or another diagnosis. Neuroimaging findings are frequently nonspecific in nature, necessitating interpretation within the context of clinical findings.20,21
Aspergillosis. The most prevalent causative species of aspergillosis is Aspergillus fumigatus. The infection is typically transmitted directly from the paranasal sinuses or from extrinsic foci, such as the lungs, through hematogenous spread. Aspergillus has been demonstrated to induce cerebritis, which may result in abscess formation (Figure 6).

Figure 6. Typical neuroimaging findings in central nervous system aspergillosis. An axial noncontrast T1-weighted image demonstrates hypointensity in the right frontal and bilateral occipital periventricular and deep white matter (A). An axial postcontrast T1-weighted image shows no significant enhancement (B). A T2-weighted image demonstrates moderate to high signal intensity consistent with vasogenic edema (C). Diffusion-weighted imaging (DWI) shows restricted diffusion in the indicated regions (D). The apparent diffusion coefficient (ADC) map confirms corresponding low signal (E). A fluid-attenuated inversion recovery (FLAIR) image demonstrates concordant hyperintensity in the affected white matter (F).
Contrast-enhanced MRI is the preferred imaging modality for evaluating parenchymal involvement; vascular imaging with magnetic resonance angiography or CT angiography should be considered when clinical presentation or imaging findings suggest angioinvasive disease.21 These lesions are characterized by a target pattern on T2, exhibiting a central hyperintensity surrounded by a hypointense rim, attributable to hemorrhage and hyphae. The presentation of an aspergilloma may be characterized as either a nodular enhancing lesion or a rim-enhancing lesion. In the angioinvasive form of the disease, vascular complications may occur, including vasculitis, mycotic aneurysms, and ischemic or hemorrhagic strokes.21,22
Cryptococcosis. Cryptococcosis, caused by Cryptococcus neoformans, is a major opportunistic CNS infection in people with AIDS.21,22 In most cases, hematogenous spread is observed from a pulmonary infection. The most frequent manifestation is meningitis, characterized by the presence of leptomeningeal enhancement. Typically, the basal ganglia, thalamus, and subcortical white matter become involved through perivascular spread. Involvement of dilated perivascular spaces is characterized by hypodensity on CT, T1-weighted isointensity or hypointensity, and T2-weighted and FLAIR hyperintensity. These lesions usually manifest bilaterally in the basal ganglia, with progression to confluent cystic lesions (also termed gelatinous pseudocysts) with mild mass effect, enhancement, and restricted diffusion. Cryptococcomas—chronic granulomas that present as a result of direct invasion of the brain’s parenchyma—are characterized by T1-hypointense and T2-hyperintense lesions. When the ependymal surface is involved, communicating hydrocephalus may develop.14,21-23
Parasitic Infections
Neuroparasitic infections are caused by a wide variety of organisms capable of affecting both immunocompetent and immunocompromised individuals.7,10
Toxoplasmosis. Toxoplasmosis, caused by Toxoplasma gondii, is the most common neuroparasitic infection among people with AIDS.14,24 Congenital toxoplasmosis has a tendency to affect the brain and eyes.14,24 Multiple lesions are predominantly observed in the basal ganglia, thalamus, and corticomedullary junction. Typical cases demonstrate T1 hypointensity and T2 hyperintensity with a DWI signal that usually is hypointense or isointense. A characteristic peripheral rim, which is hyperintense on T1 and hypointense on T2 and demonstrates restricted diffusion, is commonly seen. Areas of susceptibility are observed due to the presence of hemorrhage. After administration of contrast medium, ring or nodular enhancement is observed. An enhancing eccentric nodule gives rise to the characteristic eccentric target sign (Figure 7).

Figure 7. Neuroimaging findings associated with cerebral toxoplasmosis in an individual with AIDS. Diffusion-weighted imaging demonstrates multiple lesions with relatively decreased central diffusion signal and a peripheral rim pattern (arrows) (A). A T2-weighted/fluid-attenuated inversion recovery image demonstrates a large left basal ganglia lesion with marked surrounding vasogenic edema (B). An axial noncontrast T1-weighted image demonstrates multiple lesions (C). An axial postcontrast T1-weighted image shows multiple ring-enhancing lesions (arrows), consistent with toxoplasmosis (D).
Neurocysticercosis. Neurocysticercosis is caused by the tapeworm Taenia solium. Cysticercal cysts are predominantly localized in the cerebral cortex, basal ganglia, and subarachnoid spaces. Multilocular extra-axial cysts are known to form a grape-like formation in the CSF spaces. The most prevalent clinical manifestations include headache and seizures.
The infection progresses through 4 distinct stages. In the vesicular stage, the cyst appears T2 hyperintense and contains an eccentric T2 hypointense focus, without enhancement. In the colloidal stage, the cyst typically demonstrates ring enhancement accompanied by perifocal edema. In the granulomatous (degenerative) stage, calcification begins to appear, and both enhancement and edema diminish. In the nodular calcified stage, neither enhancement nor edema is present. Differentiating the colloidal and granulomatous stages from metastases or abscesses is often challenging, largely due to overlapping ring-enhancing appearances and surrounding edema; however, the presence of an eccentric scolex, lesion multiplicity, and lack of marked diffusion restriction favor neurocysticercosis.14,21,24
Spirochetal Infections
Lyme Neuroborreliosis. Lyme disease (ie, Lyme borreliosis) is an infection caused by spirochetes of the Borrelia burgdorferi sensu lato complex, transmitted to humans primarily through the bite of infected ixodid ticks. As a multisystem disease, Lyme borreliosis can involve the nervous system—a presentation termed Lyme neuroborreliosis (LNB). Neurologic involvement typically occurs weeks to a few months after infection. LNB may manifest in a wide spectrum of clinical presentations, including radiculoneuritis, lymphocytic meningitis or meningoradiculitis (Bannwarth syndrome), myelitis, unilateral or bilateral facial nerve palsy, other cranial nerve palsies, encephalitis, peripheral neuropathy, cerebral vasculitis, or optic neuritis.25
The diagnosis of LNB is primarily based on suggestive neurologic symptoms, CSF pleocytosis, and evidence of intrathecal synthesis of B burgdorferi–specific antibodies. Although neuroimaging findings are not diagnostic, characteristic imaging patterns may support the diagnosis and help exclude alternative causes. MRI is the preferred modality over CT for the evaluation of neuroborreliosis.25
Cranial neuritis is characterized by cranial nerve root enhancement on gadolinium contrast-enhanced T1-weighted imaging, most commonly affecting cranial nerve VII (bilaterally in ~25% of cases), with less frequent involvement of cranial nerves VIII, V, IV, and III (Figure 8). Meningitis manifests as leptomeningeal enhancement on postcontrast T1-weighted imaging, which may rarely extend along the spinal meninges. Encephalitis typically presents with small, nonspecific T2-hyperintense foci in the periventricular, subcortical, or brainstem regions that usually do not enhance; less common patterns include larger tumefactive T2-weighted lesions.26

Figure 8. Lyme neuroborreliosis with meningitis and cranial polyneuritis in an individual, age early 30 years, presenting with progressive headache, fever, diplopia, right facial nerve palsy, and partial right abducens palsy. Cerebrospinal fluid analysis showed lymphocytic pleocytosis, and Lyme serology was positive, with clinical improvement after treatment with ceftriaxone and doxycycline. An axial noncontrast T1-weighted image is presented (A). Axial postcontrast T1-weighted images demonstrate enhancement of the right trigeminal nerve and facial nerve, consistent with cranial neuritis (B and C).
Prion Disease (Sporadic Creutzfeldt-Jakob Disease)
Creutzfeldt-Jakob disease (CJD) is a spongiform encephalopathy and prion disease. The characteristic clinical presentation of CJD is rapid neuropsychiatric decline that may manifest as dementia, behavioral abnormalities, aphasia, apraxia, or frontal lobe syndromes.
CJD is best identified by MRI evaluation, which can be pathognomonic in the proper clinical setting. DWI has shown a reported sensitivity of 91% and specificity of 97% when obtained early in the disease course.27 CJD presents with evolving symmetric regions of high signal on DWI and FLAIR within the cortical ribbon, thalami, and basal ganglia. In the absence of a recent history of status epilepticus, anoxic encephalopathy, or acute viral encephalitis—disorders that can have a similar appearance on MRI—these imaging changes are considered pathognomonic. Figure 9 depicts imaging results typical of sporadic CJD, demonstrating the typical bright symmetric DWI signal within the thalami and throughout the cortical ribbon. In spite of imaging features that should be readily apparent to all neuroimaging specialists, the diagnosis is missed in two-thirds of radiology reports.28

Figure 9. Diffusion-weighted imaging in sporadic Creutzfeldt-Jakob disease demonstrates hyperintensity in the pulvinar nuclei, corresponding to the pulvinar and “hockey stick” signs (arrows) (A). Additional increased diffusion-weighted imaging signal is present throughout the cortical ribbon (arrow) (B).
Conclusion
CNS infections are time-critical conditions in which neuroimaging directly informs diagnosis, triage, and management. In routine clinical practice, a structured, indication-driven approach based on the clinical presentation (eg, meningitis, encephalitis, focal lesions), immune status, and suspected complications guides the selection of appropriate imaging modalities and sequences, thereby reducing diagnostic delays.
CT is valuable for initial emergency assessment (eg, hemorrhage, hydrocephalus, mass effect) and procedural decision-making, but MRI is preferred for defining parenchymal, meningeal, ventricular, and vascular involvement, and for follow-up. Across entities, a small set of MRI components provides the highest yield: DWI with ADC mapping (to identify purulent material and acute infarction), contrast-enhanced T1-weighted imaging and postcontrast FLAIR (to improve detection of leptomeningeal disease and characterize ring-enhancing lesions), and SWI (to detect hemorrhage in angioinvasive infections). Recognizing pattern-based combinations, such as restricted diffusion in pyogenic abscesses, basilar leptomeningeal enhancement with hydrocephalus in tuberculous meningitis, and limbic predilection in HSV encephalitis, supports timely antimicrobial therapy, may help identify individuals who might require neurosurgical intervention or critical care, and could guide monitoring for complications. Because imaging appearances may overlap with autoimmune, neoplastic, or vascular mimics and may be subtle in early disease, neuroimaging findings must always be interpreted in close correlation with clinical presentation, cerebrospinal fluid analysis, and microbiologic data, particularly in immunocompromised individuals.
Ready to Claim Your Credits?
You have attempts to pass this post-test. Take your time and review carefully before submitting.
Good luck!
Recommended
- Imaging & Testing
Neuroimaging After Brain Radiotherapy and Radiosurgery
Venkatesh Shankar Madhugiri, MBBS, MCh, FACS; Neil D. Almeida, MDVenkatesh Shankar Madhugiri, MBBS, MCh, FACS; Neil D. Almeida, MD - Imaging & Testing
Artificial Intelligence in Clinical Neurology: Opportunities, Limitations, and the Path Forward
Aysha Jadran, MD; Saqib A. Chaudhry, MDAysha Jadran, MD; Saqib A. Chaudhry, MD



