First-In-Human Study Results Show PTI-125, An Investigational Agent, Reduces Biomarkers of Alzheimer’s Disease, Inflammation, and Neurodegeneration
At the Clinical Trials in Alzheimer Disease meeting in San Diego, CA December 4-7, 2019, results of an open label first-in-human clinical trial (NCT03748706) of PTI-125 (Cassava Sciences, Austin, TX) were presented. An investigational agent, PTI-125 is being investigated for potential treatment of Alzheimer’s disease (AD). The results showed that PTI-125 reduced neuroinflammatory markers and markers of neurodegeneration.
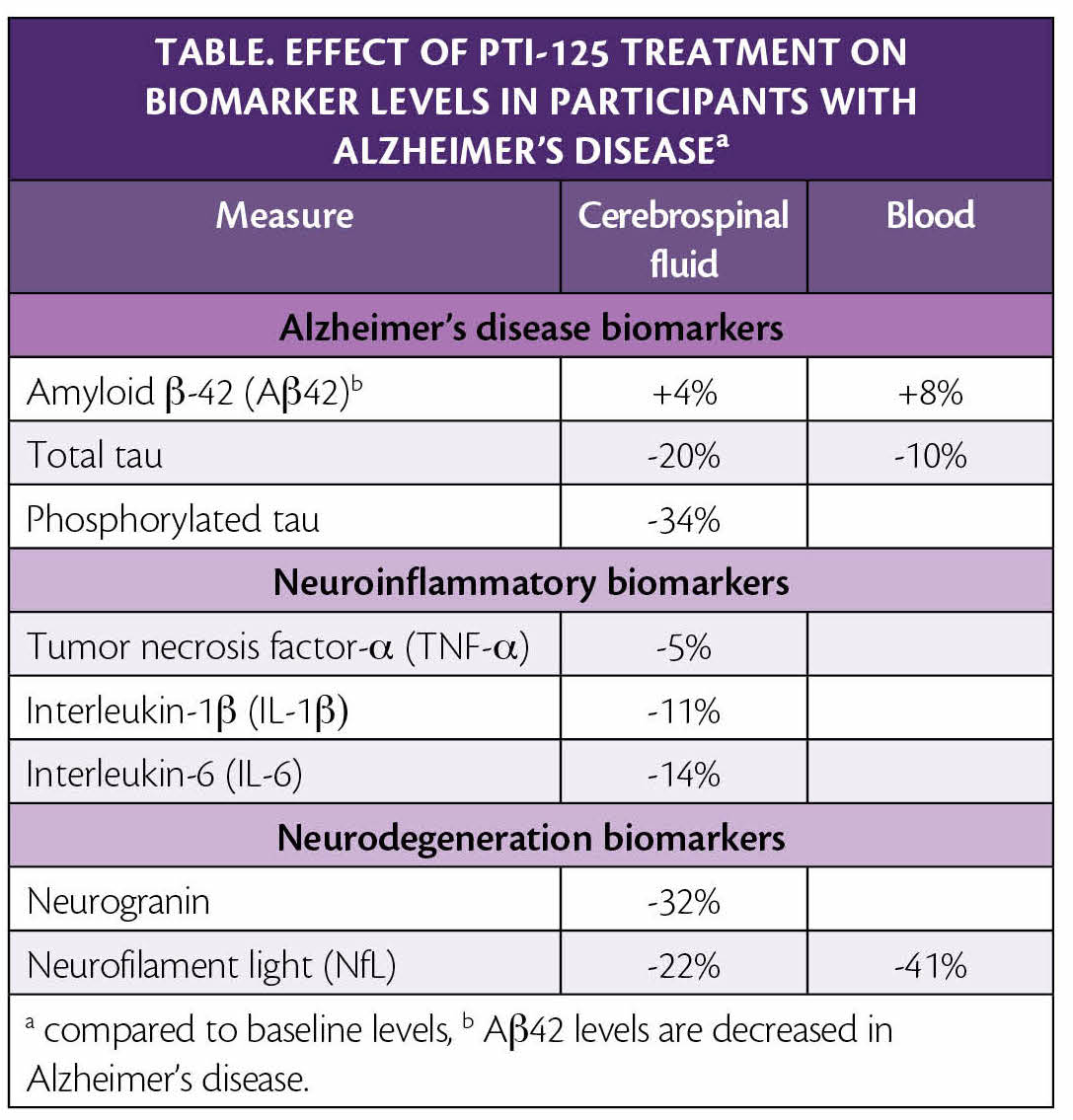
Individuals with mild to moderate AD, age 50 to 85 with Mini-Mental State Examination (MMSE) scores between 16 and 24 were treated with 100 mg of PTI-125 (100 mg twice daily) for 28 days. No drug-related adverse events occurred. Reductions from baseline were seen in cerebrospinal fluid (CSF) levels of AD biomarkers, inflammatory biomarkers, and neurodegeneration biomarkers (Table).
An ongoing blinded placebo-controlled randomized clinical trial in a cohort of 60 participants is underway with the aim of confirming these results.
“These results are exciting as it further demonstrates the clinical benefits and importance of slowing down neurodegeneration and neuroinflammation in Alzheimer’s patients. A 100% response rate in a small phase 2a study is promising in our hopes to confirm the novel drug's effects on all biomarkers tested in the phase 2b study,” said Cassava Sciences’ chief executive officer Remi Barbier.
A small molecule, PTI-125 binds altered filamin A (FNLA) found in the brains of people with AD. In its unaltered form, FNLA is a neuronal membrane protein that acts as an intracellular scaffolding protein. Altered FNLA enables amyloid ß-42 (Aß42) signaling at the acetylcholine receptor that results in tau phosphorylation and at the toll-like receptor 4 (TLR4) to result in release of inflammatory cytokines. It is thought that by binding to altered FNLA, PTI-125 reduces phosphorylated tau and inflammatory cytokines, inhibited the inflammation and neurodegeneration that are hallmarks of AD.
