Blood Biomarker May Predict Concussion Severity and Duration
According to a study published in the July 8, 2020 issue of Neurology, serum levels of the biomarker neurofilament light chain (NfL) can accurately predict the severity of the injury and how long it will last in individuals who have had concussions (mild traumatic brain injuries [mTBI]) as well as levels in cerebrospinal fluid (CSF).
The amount of the biomarker in the athletes’ blood was similar to the level in their spinal fluid. Biomarker levels were different in participants who had no concussions, concussions less than a year before, or concussions more than a year before.
“When your brain is injured, neurofilament light chain levels are higher in both your blood and your spinal fluid,” said study author Pashtun Shahim, MD, PhD, of the National Institutes of Health Clinical Center in Bethesda, Md. “Measuring this biomarker in your blood with a simple blood draw is faster and easier than measuring it your spinal fluid, which requires a more invasive lumbar puncture. Our findings are exciting because they show that the simple test may also be just as accurate for determining how severe the injury is and predicting how you might do long term.”
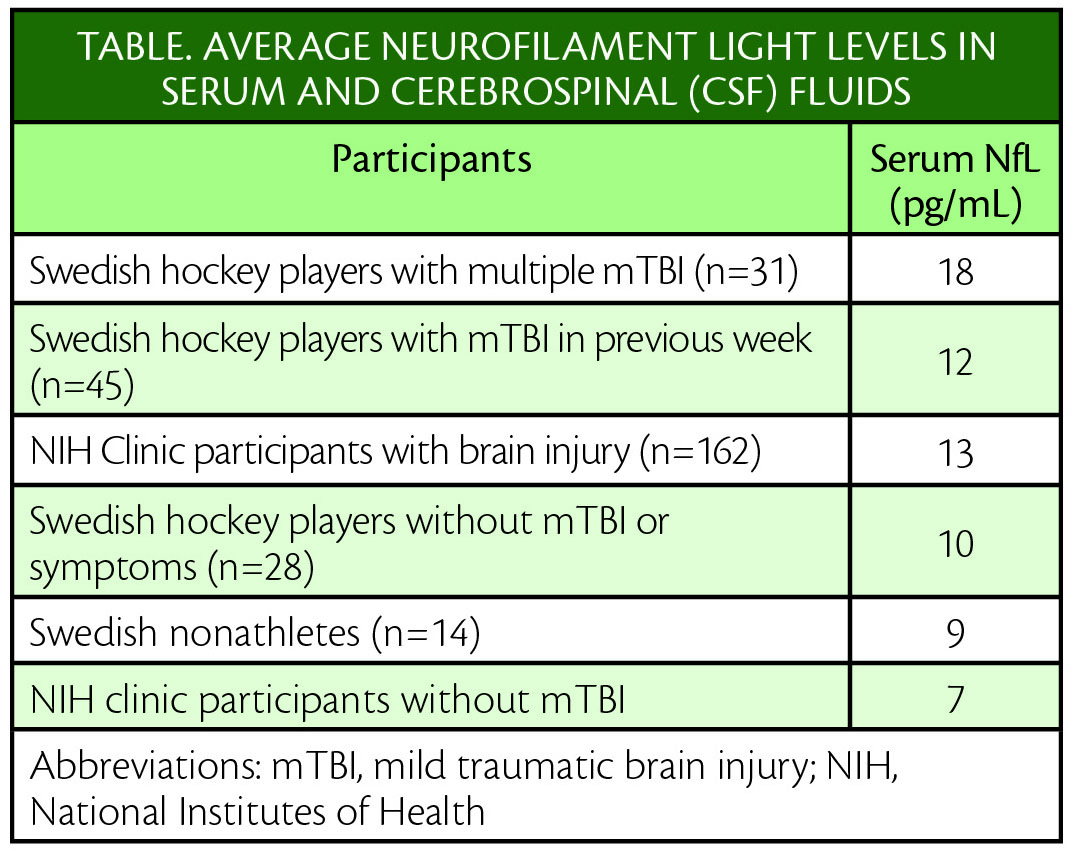
The study involved 2 groups of individuals with head injuries. The first group consisted of 104 professional Swedish hockey players ( median age 27) and included 45 athletes ith mTBI within the past week, 31 with who had multiple mTBIs, 28 athletes with no recent mTBI or symptoms, and 14 nonathletes. The second group, based at the National Institutes of Health Clinical Center, had an average age of 43 and included 162 people with brain injuries and 68 healthy people.
All of the participants had the biomarker levels in their blood tested. The 31 athletes with multiple mTBI and the 14 nonathletes also had the biomarkers in their spinal fluid tested through spinal taps and levels correlated between serum and CSF.
