Epilepsy Essentials: VideoEEG Monitoring
The development of digital video and EEG recordings has expanded the utility of the procedure beyond epilepsy monitoring but has also introduced many complex issues. These complexities relate to appropriate clinical indications, acceptable patient safety, and proper recording environment, equipment, and techniques. This first of a 2-part special report addresses methods to optimize recording techniques and interpret video and seizure semiology from videoEEG monitoring. In the next issue of Practical Neurology, the second part will address how to interpret EEG data from videoEEG monitoring in adults. Most of the information covered in both parts of this special report is equally applicable to pediatric patients, especially school-age children and adolescents.
Premonitoring Evaluation and Preparation
Although the usefulness of describing seizures and seizure-like events has been doubted,1-4 the neurologic history enables clinicians to decide whether there is an indication for videoEEG monitoring. Because the basis for spells is obvious from the initial clinical evaluation in some individuals, not all paroxysmal events require videoEEG monitoring, making it possible to avoid the considerable expense and inconvenience associated with videoEEG procedures. In others, videoEEG monitoring may not be cost-effective because their spells are infrequent, unless the timing of spells is relatively predictable, or the spell occurrence can be enhanced by medication withdrawal or reliable provocative factors. During history taking, factors that precipitate seizures should be identified so these may be duplicated during videoEEG monitoring to enhance the occurrence of spells. The list of seizure and seizure-like event descriptors in Checklist 1 should all be described and analyzed before considering videoEEG monitoring.
Management During Monitoring
Admission, Medication Management & Sleep Deprivation
We attempt to duplicate factors that have been reported by patients to be potentially aggravating for their seizures. Although some studies have cast doubt on the value of sleep deprivation in the monitoring setting,5 sleep deprivation is one of the most frequent reasons for seizure aggravation in the everyday living situation.6-8 Other provocative tests (eg, photic stimulation, exercise, or hyperventilation) should also be considered.
It is appropriate to begin withdrawing antiseizure medications (ASMs) before the day of admission when habitual spells are not intense and when they have not responded to ASM treatment. As a rule, we do not withdraw ASMs until the morning of admission because the inpatient setting is a safer environment in which to do so.9 In general, ASMs are tapered daily at the rate of one-third of the original dose. Medications that are pharmacokinetically or pharmacodynamically long acting may be discontinued (eg, zonisamide, phenobarbital, valproate, and clobazam) but note that concomitant use of enzyme-inducing drugs may shorten the half-life. Occasionally, ASM discontinuance or tapering may result in “withdrawal” symptoms such as restlessness, insomnia, and dysphoria. Withdrawal is more common with barbiturates and benzodiazepines but may also occur with carbamazepine.10 Diazepam or lorazepam are effective for sedation to address these symptoms but may impede seizure recording. If needed, 25 mg of oral nortriptyline or 25 to 50 mg of diphenhydramine is effective in controlling withdrawal symptoms (especially with carbamazepine) and typically does not reduce the chances of recording seizures.
Rapid ASM tapering (daily dose reduction of 30%-50%) has been shown to significantly reduce length of stay, without any serious adverse events in people with drug-resistant epilepsy.11,12 ASM withdrawal could, however, contribute to the precipitation of generalized tonic-clonic seizures (GTCS), status epilepticus (SE), and the more feared sequelae of sudden unexpected death in epilepsy (SUDEP).13 SUDEP after a GTCS has been observed, typically following a 50% to 100% ASM withdrawal.13 In studies comparing rapid vs slow tapering of ASMs, SE was not reported, although persons with a history of SE were excluded from these studies.11,14 In lieu of a fixed ASM taper protocol, each patient should be assessed individually regarding the safety of ASM withdrawal.
Guidelines for treating prolonged or frequent seizures are needed and should strike a balance between the need to record further spells and initiating the “emergency” or “rescue” plan for safety. Typical but not routine parameters for administering parenteral medications include: 1) 2 GTCS within 2 hours; 2) generalized convulsive activity lasting more than 5 minutes; 3) 6 focal impaired-awareness seizures within 24 hours; or 4) a focal seizure lasting more than 10 minutes. Patient safety is the primary concern, and parameters need to be modified according to the individual patient’s health, seizure type, and epilepsy history.15 In some, history may indicate the point at which seizures are likely to become prolonged or clustered. Oral treatment may be more appropriate than intravenous ASMs for less urgent instances. Sustained or continuous release forms of ASMs should not be used for the purpose of orally loading ASMs.
General Safety in the Adult Epilepsy Monitoring Unit
In the general population, people with epilepsy have an increased risk of injury from their seizures. Despite best efforts, adverse events can occur in the epilepsy monitoring unit (EMU) with adverse events requiring intervention reported in up to 21% of patients.15 Over 50% of adult EMUs in North America report falls or postictal psychosis in a 12-month period.16 Other reported complications include psychiatric adverse events, aspiration, and cannula-related thrombophlebitis.17 Routine safety procedures are described in Checklist 2.12
Exercise programs are implemented to prevent physical deconditioning due to bed rest. The combination of venous thromboembolic prophylaxis (eg, heparin or low molecular weight heparin) and physical activity reduces the risk of deep vein thrombosis. Falls are a frequently reported complication,13 and we use a ‘walking sling’ (Figure), allowing safe ambulation with a mobile harness attached to the ceiling via a track. Persons being monitored are encouraged to use a recumbent bicycle or treadmill for exercise. While in bed, both padded guardrails and a safety belt anchored to the bed are used to prevent falls during a seizure or in sleep (Figure).
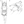
Psychiatric adverse events have been reported in 5% of adults monitored in EMUs, and over half of surveyed adult EMUs.16,17 Postictal psychosis is the most frequent manifestation, and many who experience it require formal hospitalization.17 Postictal psychosis can occur during or after the videoEEG monitoring period. A personal history of psychiatric comorbidities increases the risk of psychiatric adverse events (16-fold), and patients should be counseled about risk of exacerbation prior to EMU admission.17 Mental state should be assessed daily on rounds, and it may be necessary to involve psychiatry specialists and consider using antidepressant or antipsychotic medications during an adult EMU stay.
SUDEP has been reported to occur at a rate of 1.2 (0.6-2.1) per 10,000 monitoring sessions,13 which is thought to be similar to the out-of-hospital rates in people with drug-resistant epilepsy.13 Our EMU has 24-hour on-site video, EEG and ECG monitoring by specialty-trained technicians who are in close contact with ward nursing staff, and on-call epilepsy physicians. Live visual EEG monitoring by the technologist is sometimes supplemented by real-time seizure and spike detection software. When a clinically relevant event is noted, the technologist alerts the nursing staff, and they attend to the patient with airway and cardiac assessment as discussed earlier. In some cases, SUDEP was thought to have been averted by the prompt cardiorespiratory support by staff.
Spell Evaluation
Analysis of Video Data and Peri-ictal Testing
Peri-ictal (ictal and postictal) examination is important to elicit semiologic features of seizures that otherwise would not be apparent. Attempts should be made to test for:
- response to communication,
- response to physical stimulation,
- >memory, by presenting phrases or words for later recall,
- distractibility,
- weakness or lack of motor control,
- response to passive eye opening,
- plantar extensor response, and
- speech (naming, reading)
Of the many possible types of spells in adults, epileptic seizures, psychogenic nonepilepstic seizures (PNES), and syncope are most common during videoEEG studies. The 3 helpful semiologic clues to the epileptic nature of spells are: 1) localizing semiologic features; 2) physiologic sequence of progression; and 3) other peri-ictal signs of an epileptic seizure mechanism.
Localizing Semiologic Features. Observable peri-ictal semiologic features can be grouped by motor signs (positive or negative), automatisms, autonomic signs, and speech disturbance (Table 1).18 A detailed discussion of the known or presumed mechanisms underlying some of these signs is outside the scope of this article and can be found elsewhere.19,20


The temporal and frontal lobes are the 2 brain regions most frequently affected by focal epilepsy. Distinguishing between temporal and frontal lobe seizures (Table 2) is important in evaluation of drug-resistant epilepsy and whether a person is a candidate for resective epilepsy surgery. Using the video-recorded features in Table 2, we found seizures were localizable to the frontal lobe in approximately 83% of the persons with frontal lobe epilepsy, and to the temporal lobe in all persons with temporal lobe epilepsy.21

Physiologic Sequence of Progression. The physiologic sequence of how signs and symptoms progress is especially helpful in distinguishing epileptic seizures from PNES. Focal seizures with secondary generalization are an example commonly seen in adults.22 With this seizure type, the initial symptom or sign (eg, aura or focal motor activity) indicates seizure onset at a cerebral focus. Ensuing loss of awareness with automatism reflects limbic involvement, and subsequent contralateral extremity posturing is due to centripetal spread of epileptic discharge into the basal ganglia. Seizure generalization into tonic-clonic activity affects the contralateral face or extremities, followed by ipsilateral involvement. This physiologic sequence of spread of activity is lacking in PNES, in which motor signs or behaviors appear in a random sequence and distribution.
Moreover, the motor or behavioral activity often occurs in bursts, rather than unfolding in a sequential manner as is seen in epileptic seizures. Features that are useful in distinguishing PNES from epileptic seizures include a lingering prodrome, out-of-phase convulsive activity, nonphysiologic spread, pelvic thrusting, side-to-side head or limb shaking, opisthotonus, eyes and mouth shut tight, geotrophic eye deviation, bursting “stop-go” (“reprisal phenomenon”), irregular progression, distractibility, abrupt cessation, disproportionate postictal mental status, feeble motor response or vocalization (whispering), stuttering speech,20 long duration of spells, tongue bitten at the tip, normal breathing or hyperventilation immediately after cessation of convulsion, and lack of stereotypy between spells.
Note that this list does not include “positive provocative test,” which is the ability to induce a spell through suggestion. The reason is that as many as 15% of patients with epilepsy can be induced with intravenous (IV) saline into having de novo PNES,24 even with relatively mild suggestion, such as telling patients the injection “occasionally precipitated seizures in patients with seizure tendencies.” If stronger suggestions are made,25 the false positive rate for PNES is expected to be even higher. This potential confusion can lead to disastrous consequences if a clinician mistakes the presence of PNES as a negative indicator for epilepsy and withdraws ASMs. Thus, it is very important that the clinician confirms that there are no epileptic or other types of seizures that need to be evaluated. Provocative tests must be performed in the setting of videoEEG recording. The recorded event needs to be shown to the patient’s family or friends to confirm that it is the same as habitual spells, and that no other type of spell is in question.
Other Peri-ictal Signs of Epileptic Seizure Mechanisms. The following are helpful in reinforcing a suspicion for epileptic seizures rather than psychogenic spells: seizures arising during sleep, lateral tongue bite, physical injury, postictal Babinski sign, ictal cry, stertorous breathing, or foaming at the mouth.26-33
Summary
VideoEEG monitoring is a powerful diagnostic tool when utilized in an appropriate patient population with a focus on patient safety. Systematic planning and clear protocols and an individualized patient-centered approach are required. In part 2 of this special report, we will discuss analysis of videoEEG data and discharge management.
Ready to Claim Your Credits?
You have attempts to pass this post-test. Take your time and review carefully before submitting.
Good luck!
Recommended
- Epilepsy & Seizures
Functional Neurologic Symptom Disorder Resolving After Endoscopic Encephalocele Repair
Charles F. Palmer, MD; Rodney J. Schlosser, MDCharles F. Palmer, MD; Rodney J. Schlosser, MD - Epilepsy & Seizures
Immunotherapy for Management of Refractory and Super-Refractory Status Epilepticus
Eugenia Ho, MD; Mackenzie Silverman, MDEugenia Ho, MD; Mackenzie Silverman, MD - Imaging & Testing
Neuroimaging: Clinical Impact of Imaging Findings
Laszlo Mechtler, MD, FAAN, FEAN, FASN, FAHSLaszlo Mechtler, MD, FAAN, FEAN, FASN, FAHS



