Tuberous Sclerosis Complex
Pediatric epilepsy syndromes, or epilepsy syndromes in general, have traditionally been defined as a collection of clinical signs and symptoms that co-occur in a known pattern. Such patterns include having specific seizure types, along with characteristic findings on EEG, neuroimaging, laboratory testing, or involvement of other organ systems. For example, in tuberous sclerosis complex (TSC), there may be dermatologic signs (eg, ash leaf hypopigmented spots or facial angiofibromas); kidney, heart, eye, and sometimes, lung involvement; and neurodevelopmental concerns. As more pathogenic physiologic pathways are understood, as in the identification of TSC1 and TSC2 variants contributing to the function of the mechanistic target of rapamycin (mTOR) pathway,1,2 the definition of many epilepsy syndromes includes identifying the presence of such variants, which can also provide a more precise diagnosis.
Identifying the clinical pattern and, when possible, a pathogenic gene variant can give a clear diagnosis for an often difficult-to-manage set of symptoms, which has been shown in other epilepsy syndromes and neurodegenerative diseases to be helpful to patients, families, and care partners.3,4 Importantly, arriving at a clear diagnosis as early as possible can also guide treatments and provide prognostic information. Because not all symptoms occur at the same time of life in epilepsy syndromes, early diagnosis can also guide monitoring for anticipated clinical problems, leading to earlier care for those issues.
TSC is an autosomal dominant neurocutaneous epilepsy syndrome that was well defined before a specific causative gene variant was discovered.5 This brief review covers the diagnosis and management of TSC for those not specializing in pediatric epilepsy syndromes to guide them in caring for patients with TSC—which ideally should be done in collaboration with a multidisciplinary team at a tertiary-care epilepsy center.
The Genetic Basis of TSC
In the 1990s, multilinkage analysis of families that had multiple generations of people with TSC led to the identification of the TSC2 and TSC1 genes,1 which respectively encode the proteins hamartin and tuberin. These proteins form the tumor suppressor complex mTORC1. The mTOR pathway mediates cell proliferation, migration, and programmed cell death and has a role in tumorigenesis.6 Pathogenic variants of TSC1 are more likely to be familial (ie, germline mutations), whereas pathogenic variants of TSC2 are more sporadic.2 Genetic testing of individuals and their family members, when available, may lead to further understanding of genotype-phenotype correlations and disease mechanisms. Such knowledge will hopefully continue to increase the availability of treatments to address symptoms and slow or stop disease progression.
TSC Clinical Presentation
TSC classically affects the brain and skin, as well as many other body systems, and 75% to 90% of people with TSC will have seizures that are often resistant to pharmacotherapy.
Neurologic and Ophthalmologic Manifestations
Infantile spasms are often the first reason for presentation of a child with TSC to neurology. These seizures typically have onset between age 4 to 7 months and appear as sudden, brief stiffening of the muscles or half or all the body with the head thrown back. Infantile spasms frequently occur during waking from sleep. On EEG, infantile spasms are characterized by poorly organized interictal (ie, between seizures) and background hypsarrhythmia, which is random or chaotic high-amplitude slow waves with intermixed multifocal spikes. Other seizure types usually develop over time in those with TSC, especially focal impaired awareness and focal motor seizures.
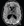
Brain MRI shows cortical tubers in up to 90% of affected individuals (Figure). Focal cortical dysplasia and cerebral white matter radial migration lines are also seen. Up to 80% of cases also have subependymal nodules along the lateral and third ventricles, sometimes described as appearing like dripping candle wax. These nodules can transform into subependymal giant cell astrocytomas (SEGAs; 15% of cases), which increases risk of obstructive hydrocephalus. Subependymal nodules typically do not occur after age 20.7
Neurodevelopmental conditions, including autism and intellectual disabilities, are frequent in TSC and the TSC-associated neuropsychiatric disorders (TAND) checklist (Table 1) may help identify these manifestations.8 Identification of neurodevelopmental and neuropsychiatric conditions is paramount as these impact daily quality of life. Additionally, intellectual disability in this population has been suggested to be associated with a higher risk of premature death from SEGAs, kidney manifestations (see Internal Organ Manifestations), or sudden unexpected death in epilepsy (SUDEP).9

Fundoscopic eye examination may reveal retinal hypopigmented patches or retinal hamartomas, the latter of which have histologic features similar to the cortical tubers found in the brain. Retinal hamartomas are a highly specific diagnostic feature but are unlikely to affect vision.10
Skin Manifestations
Small hypopigmented areas on the skin, termed ash leaf spots, are often present at birth or in early infancy and occur in approximately 90% of cases.7 Other skin findings include facial angiofibroma and fibrous cephalic raised or bumpy areas, which when found on the lower back or flank are termed Shagreen patches. Although fibrous plaques and Shagreen patches are less common (~25% of cases), they are more specific for TSC.7 Nail fibromas, dental pits, and intraoral fibromas may later develop, usually in adulthood, and can be a source of infections and irritations requiring treatment.
Internal Organ Manifestations
Cardiac rhabdomyomas, which are benign tumors, may be visible in the heart on prenatal ultrasound or shortly after birth on echocardiography. Frequently found in the heart ventricles, rhabdomyomas may have no clinical effects or may obstruct blood flow. Rhabdomyomas typically involute such that they may no longer be visible on echocardiography with time but can still cause arrhythmias.11 When present prenatally or neonatally, cardiac rhabdomyomas can be a signal to evaluate family history, initiate genetic counseling for the family as well as genetic testing, and plan further monitoring of all potentially impacted organ systems for the affected child. Often there is a family history, but TSC can arise from a de novo mutation.12
Renal angiomyolipomas (AMLs) are benign, heterogeneous tumors frequently occurring in TSC. AMLs often bleed as they grow. Multiple renal cysts may be present, more so with specific genotypes.12 Renal involvement in TSC may make dialysis or kidney transplant necessary and can be a cause of premature death. AMLs rarely present in organs other than the kidney.
Smooth muscle cell infiltration of the lung, manifesting in pulmonary lymphangioleiomyomatosis (LAM), may occur in the third or fourth decade of life, presenting as progressive difficulty breathing, bloody cough, and lung collapse. LAM has been reported more often in women (≤40%) and rarely in men with TSC. Sporadic LAM with or without renal AMLs may occur without other features of TSC due to somatic gene mutations. Individuals with lung involvement may be at higher risk of death within a few years of pulmonary symptom onset.
TSC Diagnosis and Monitoring
Clinical recognition of TSC is strong enough that genetic testing is often confirmatory only. Typically, diagnosis is made with imaging studies of the brain, heart, and eyes and dermatologic evaluation after a child presents with infantile spasms, although as noted, prenatal detection of cardiac rhabdomyomas may also lead to diagnosis. Family history should be taken and skin examination and neuroimaging done for any child with infantile spasms because these may be key in making a diagnosis of TSC and guiding surveillance of other body systems. Early diagnosis has become more paramount as treatments targeting the mTOR pathway have become available. Diagnostic criteria are provided in Table 2.

Because of the multisystem nature of TSC and the lifelong course with new symptoms appearing over time, surveillance monitoring is essential. Recommendations for monitoring are provided in Table 3.11

TSC Treatment
Recent evidence suggests that early pharmacologic intervention, possibly before seizure onset, may reduce the severity of both epilepsy and neuropsychiatric manifestations of TSC.13-15 The timing of intervention may relate to the development of epileptiform abnormalities on screening EEG in infants with TSC even prior to clinical seizures. For patients with TSC who have not developed seizures, their families should be counseled on seizure recognition, especially as infantile spams can be subtle events clinically. Antiseizure medications (ASMs), epilepsy surgery, dietary modification, and treatment with mTOR inhibitors are options to consider for TSC.14 Importantly, therapies for neuropsychiatric comorbidities and interventions to improve quality of life are also available.
ASMs
Vigabatrin is a first-line treatment for infantile spasms in TSC and should be started as soon as this seizure type is confirmed. Most infantile spasms are significantly reduced or even stopped with vigabatrin. There is a risk of visual impairments when vigabatrin is taken over prolonged periods; however, this is uncommon with treatment for the duration of time when infantile spasms occur.16 If sufficient response does not occur with vigabatrin, prednisone/prednisolone or adrenocorticotropic hormone (ACTH) may be beneficial.17
Everolimus is an mTOR inhibitor approved for treatment of TSC-associated focal (partial-onset) seizures. In clinical trials, children with TSC treated adjunctively with everolimus had 29% to 40% reductions in seizure frequency.18,19 The starting dose is 5 mg/m2/day, which may be increased in increments of 5 mg to reach trough concentrations of 5 to 15 ng/mL. Regular laboratory monitoring is required including blood counts, renal function, liver function, lipid profile and glucose.
Cannabidiol is also approved for the treatment of seizures in TSC. In clinical trials, participants with TSC treated with cannabidiol had a median 23% reduction in seizures from baseline relative to placebo.20 The recommended starting dose is 2.5 mg/kg orally twice daily, which may be increased weekly in increments of 2.5 mg/kg twice daily to a recommended maintenance dose of 12.5 mg/kg twice daily. Lower doses should be used in patients with hepatic insufficiency and those who are treated concomitantly with everolimus; interactions with other antiseizure medications can contribute to toxicity. Regular laboratory monitoring of liver function is required.
Other ASMs commonly used in TSC include clobazam, lamotrigine, lacosamide, levetiracetam, oxcarbazepine, perampanel, topiramate, valproic acid, brivaracetam, and carbamazepine.21 Most seizure types, other than infantile spasms, in TSC are not responsive to pharmacotherapy and polypharmacy is common. Vagus nerve stimulation and the use of a ketogenic diet may be considered for pharmacoresistant seizures. Epilepsy surgery, discussed further below, may be indicated.
mTOR Inhibitors
The mTOR inhibitors sirolimus and everolimus, developed initially as immunosuppressants, have clinical value for aspects of TSC. A topical gel formulation of sirolimus (0.2%) has been approved by the Food and Drug Administration (FDA) as of April 2022 for treatment of facial angiofibromas in TSC in those age 6 years or more. Everolimus was shown in clinical trials to reduce the volume of SEGAs22 and is approved for the treatment of SEGAs not amenable to surgical resection. Renal angiomyolipomas can be treated with mTOR inhibitors, as can lung tumors.23 As noted, everolimus is also used as an antiseizure medication for treatment of seizures.
Epilepsy and Other Surgery
Because the many cortical tubers of TSC represent potential for multiple epileptogenic foci, surgical resection previously had been thought to be less of an option. More recently, it has been recognized that even in patients with TSC there may be seizure freedom or palliation from resection of a particular structural lesion that is concordant with seizure description or EEG findings. Surgical resection may be beneficial, reducing or eliminating seizures for months to years.24-26 Although other foci may remain and prevent lifelong seizure freedom, even a temporary reduction in seizure frequency and severity can improve quality of life and neuropsychiatric outcomes.26 Even if seizure freedom is not long-lasting and other seizure types develop, this period of reduced or no seizures may prevent seizures from developing in other areas of the brain or may protect brain development for a time. Individuals who benefit from these surgeries may still be candidates for other interventions, including further resections, as needed later in life.
SEGAs that are symptomatic should be resected acutely, and cerebrospinal fluid (CSF) shunt diversion may be needed. For growing but otherwise asymptomatic SEGA, resective surgery or treatment with mTOR inhibitors is warranted.27
Nephrectomy for renal angiomyolipoma should be avoided in favor of embolization and corticosteroid treatment for angiomyolipomas hemorrhaging acutely. Kidney-sparing resection may also be considered.7 If lung tumors occur, lung transplant may be considered. Consider excision or laser surgery for symptomatic, rapidly changing, or disfiguring skin lesions.
Treating Comorbidities
From the time of diagnosis and across the lifespan, the TAND checklist (Table 1) can be used to evaluate those with TSC for neuropsychiatric effects and comorbidities.8 Mood disorders, autism, anxiety, and behavioral issues are common, and all can be therapeutically addressed with interventions from psychologists, psychiatrists, occupational/vocational therapists, and speech-language pathologists. In many comprehensive epilepsy centers or TSC centers, a developmental psychologist and neuropsychologist are available.7 At the center where the author practices, consultation with these professionals at least once annually is considered standard of care.
Team-Based Care and Addressing Quality of Life
The multisystem involvement of TSC, neuropsychiatric effects, and comorbidities require team-based multidisciplinary care focused on treating symptoms and improving quality of life is recommended. Because epilepsy surgery is often indicated, these patients should be referred to tertiary epilepsy care centers early in the course of their condition, where such team-based care is often most accessible. Cardiologists, nephrologists, pulmonologists, ophthalmologists, dermatologists, and other specialists should join this team as needed as clinical manifestations develop. Although it may be difficult for families to travel to these centers, even an annual visit with collaboration and teleconsultation with the referring physician is beneficial.
Social workers in epilepsy care centers can collaborate with school systems and local resources to arrange for needed accommodations in the community and at schools and other settings. Assistance arranging respite care for the family and accessing mental health services can also be provided. The TSC Alliance (www.tscalliance.org) is a valuable resource for families with TSC and for their health care providers.
Summary
Understanding the genetic pathogenesis of TSC has led to improved diagnosis and characterization of the syndrome. Most importantly, it has led to new opportunities to intervene with neuropsychiatric care, epilepsy surgery, ASMs, and mTOR inhibitors earlier than ever before. Neuroimaging, EEG, and echocardiography are critical for diagnosis, and all patients with TSC should be monitored for all clinical manifestations of TSC across their lifespan. It is both exciting and humbling to be at this frontier, which also carries an obligation to diagnose as early as possible, counsel families well, and provide treatment as opportunities for ever-earlier treatment arise.
Ready to Claim Your Credits?
You have attempts to pass this post-test. Take your time and review carefully before submitting.
Good luck!
Recommended
- Imaging & Testing
MRI Clues to Intracranial Infection: Differentiating Bacterial, Viral, Fungal, and Parasitic CNS Disease
Márk Kozák, MD; Máté Sik, MDMárk Kozák, MD; Máté Sik, MD - Alzheimer Disease & Dementias
Special Report: 2025 Neurology FDA Drug & Device Approvals & Clearances



