ICU Care for Severe Traumatic Brain Injury
Metabolic Mismatch in Severe Traumatic Brain Injury
Severe traumatic brain injury (TBI) is defined as head trauma resulting in Glasgow Coma Scale scores between 3 and 8.1 The management of severe TBI in the intensive care unit (ICU) aims to prevent secondary brain injury, which often results from metabolic mismatch, in which the demand for energy exceeds its supply (Figure 1). Adequate metabolic delivery requires a sufficient cerebral perfusion pressure (CPP). The CPP is the difference between the mean arterial pressure and the intracranial pressure (ICP). In the uninjured brain, cerebral blood flow is constant over a wide range of mean arterial pressure (Figure 2A), as the caliber of blood vessels adjusts to accommodate changes in blood pressure (Figure 2B).2 In severe TBI, the normal regulation of cerebral vasculature is disrupted, narrowing the cerebral autoregulation curve and shifting it to the right. A primary clinical goal is to identify a target CPP, based on clinical examination and neuromonitoring data. One method is to select a CPP at which the mean arterial pressure and ICP are not correlated, as this indicates preserved autoregulation (Figure 2C).3 In addition to the delivery of blood to the brain, the content of that blood matters. Hypoxemia and hypoglycemia are potential systemic sources of secondary injury after TBI.

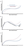
Several tools can help integrate this information to identify brain tissue metabolic conditions and causes of metabolic mismatch. Parenchymal monitors can measure ICP, oxygenation, blood flow, metabolites, and electrical activity. These monitors provide only local information at the site of probe placement; other tools, including EEG, near-infrared spectroscopy, CT angiography and perfusion, jugular bulb oxygen saturation, and transcranial doppler, can provide more global information about cerebral perfusion and energy consumption. These data, in combination with clinical examination and vital sign monitoring, can help establish an appropriate CPP target.
Airway, Breathing, and Circulation after Severe TBI
Patients with severe TBI often require intubation. Because cervical spine injury is common in these patients, the most experienced practitioner available should perform intubation, using manual inline stabilization, video laryngoscopy, and a hyperangulated blade. Anesthetic induction should aim to suppress intracranial hypertension and brain metabolic demand while not compromising systemic perfusion. The muscle relaxant succinylcholine may cause hyperkalemia in people who have experienced trauma. Therefore, rocuronium is preferred for neuromuscular blockade and can be reversed with sugammadex to restore the patient’s motor function to enable a neurologic examination to be performed that will help with diagnosis and monitoring.
Oxygenation and ventilation are also important considerations. Hypoxemia is associated with poor outcomes after TBI.4 An ongoing phase III randomized controlled trial, BOOST3 (Brain Oxygen Optimization in Severe Traumatic Brain Injury-3), is evaluating whether targeting brain tissue oxygenation improves outcomes after severe TBI.5 Emergency neurologic life support guidelines recommend targeting an oxygen saturation (SpO2) of at least 90%.6 Hypercapnia raises ICP by causing cerebral arteriolar vasodilation. Hyperventilation may reduce intracranial hypertension by inducing cerebral vasoconstriction, but prolonged hypocapnia can cause cerebral hypoperfusion and should be avoided.
The management of systemic blood pressure requires balancing the risks of insufficient and excessive perfusion. In the early period after severe TBI, especially in patients with intracranial hemorrhage, strict blood pressure control, while avoiding hypotension, reduces the risk of hematoma expansion. After hemostasis has been achieved, permissive or induced hypertension may help maintain CPP.
Intracranial Hypertension
Intracranial hypertension threatens cerebral perfusion and metabolic supply. Its treatment often begins with hyperosmolar therapy, using hypertonic saline or mannitol. These agents generate an osmotic gradient that dehydrates the brain, resulting in decreased cerebral edema and lower ICP.
Sedation and targeted temperature management7 can suppress metabolic demand, leading to cerebral vasoconstriction and reduced ICP. However, anesthetic agents confound the neurologic examination and may carry other adverse effects. Pentobarbital is highly effective at suppressing ICP but is often reserved for salvage therapy because it can cause cardiomyopathy, immunosuppression, and prolonged coma. Whereas therapeutic hypothermia has not been found to be beneficial after severe TBI,8 induced normothermia does improve ICP.7
Elevating the head promotes venous return and reduces ICP. In our center, we have developed a protocol to verticalize patients to maximize venous return and have reported on its efficacy in managing intracranial hypertension unresponsive to other treatments.9 Because verticalization can cause severe soft-tissue pressure injuries and compartment syndrome, it should be reserved for refractory cases.
Surgical interventions also may reduce intracranial hypertension. External ventricular drain placement can treat symptomatic hydrocephalus, which often occurs as a result of intraventricular hemorrhage or obstruction from mass effect. The indications for evacuation of extra-axial hemorrhages are well-established, but the case for intraparenchymal hemorrhages is more controversial.10 In one randomized controlled trial, decompressive craniectomy (bifrontal craniectomy or unilateral frontotemporoparietal craniectomy) for refractory intracranial hypertension after severe TBI resulted in lower mortality and improved average functional outcome but did not significantly increase the likelihood of a good functional outcome.11 More aggressive bifrontotemporoparietal craniectomy after diffuse TBI decreases ICP but is associated with worse functional outcome 6 months after injury.12 Brain Trauma Foundation guidelines favor decompressive hemicraniectomy in patients with refractory intracranial hypertension and focal lesions but recommend against bifrontal craniectomy.1
Other Neurologic Complications of Severe TBI
A recent study found that 12.6% of patients with moderate to severe TBI experienced seizures during their hospitalization.13 Seizure prophylaxis is recommended for the first 7 days after TBI.1 Seizures warrant aggressive treatment, but whether particular interictal EEG patterns require aggressive treatment is controversial.14 In addition, cortical spreading depolarizations—slow waves of abnormal neuronal activation—can occur after severe TBI but typically require implanted electrocorticography to detect.15 Their significance is not well-established, but they may cause secondary neurologic injury. Ketamine can suppress cortical spreading depolarizations and could be a potential future treatment.16
Cerebrovascular injury may arise after TBI from blunt traumatic injury to a blood vessel or vascular dysregulation leading to vasospasm. Blunt cerebrovascular injury may result in arterial dissection, especially of the cervical arteries. Traumatic injury of the skull, leading to venous injury and local inflammation, can cause traumatic venous thrombosis. This in turn may result in venous infarction or secondary hemorrhage. An underrecognized but common secondary phenomenon in severe TBI is cerebral vasospasm.17 Fluid resuscitation, blood pressure augmentation, and intra-arterial vasodilation may help treat symptomatic vasospasm, although data to support these therapies are sparse.
Systemic Complications of Severe TBI
Severe TBI is associated with systemic complications arising as a direct result of brain injury or as a general result of critical illness.
Paroxysmal sympathetic hyperactivity consists of episodic dysautonomia, often triggered by stimulation, with symptoms including fever, tachycardia, hypertension, diaphoresis, posturing, and agitation.18 Treatments include alpha-2 agonists, nonselective beta-blockers, the D2 dopamine receptor agonist bromocriptine, anxiolytics, and analgesics. Autonomic dysfunction after severe TBI also may lead to neurogenic stress cardiomyopathy, which classically manifests as apical hypokinesis.19 Neurogenic pulmonary edema is thought to arise from hemodynamic changes related to intracranial hypertension and an acute inflammatory response, but like other forms of pulmonary edema, is treated with diuresis.20 Acute kidney injury after severe TBI is common, usually as a result of either systemic trauma or critical illness. In addition, severe TBI often causes brain-mediated hormonal abnormalities that lead to dysregulation of sodium excretion by the kidneys. The syndrome of inappropriate antidiuretic hormone secretion and cerebral salt wasting both cause hyponatremia, whereas diabetes insipidus causes hypernatremia.
The management of bleeding and coagulopathy after severe TBI is complex. When patients present with intracranial hemorrhage, achieving hemostasis takes precedence. Pharmacologic approaches such as treatment with tranexamic acid have not shown benefit.21 Once bleeding has stabilized, it is important to start chemoprophylaxis for venous thromboembolism as soon as it is safe to do so, because the release of tissue factor from injured brain predisposes patients with TBI to venous thrombosis and pulmonary embolism.
Other common complications in critically ill patients include ventilator-associated pneumonia, urinary tract infections, bacteremia, and pressure ulcers. Timely extubation, pulmonary hygiene, avoidance of indwelling urinary catheters, judicious use of central venous catheters, and meticulous nursing care can help reduce these complications.
Prognostication in Severe TBI
An understanding of prognosis after severe TBI—aided by clinical examination, consideration of demographic characteristics, imaging findings, and systemic factors—is essential to guiding treatment decisions.22 On examination, Glasgow Coma Scale score and pupillary reactivity have prognostic value. That said, prognostication based on the examination should be cautious early in the disease course before the patient has received adequate resuscitation or emergent surgical intervention. Several CT classification systems for TBI also exist, with the Marshall and Rotterdam scales being the most commonly used. These factors are integrated into 2 similar but distinct scoring systems: 1) the International Mission on Prognosis and Analysis of Clinical Trials in Traumatic Brain Injury (IMPACT), and 2) the Corticosteroid Randomization after Significant Head Injury (CRASH) scales.
Opportunities for the Future
The diversity of injury mechanisms and manifestations of TBI has limited progress in the development of new therapies. Despite the lack of success of glucocorticoids,23 robust immune activation after TBI would suggest that immunomodulation is an attractive target for disease-modifying therapies. In addition, inhibition of the SUR1-TrpM4 nonselective cation channel by glibenclamide, currently being studied as a potential treatment for cytotoxic edema in ischemic stroke and contusion expansion in TBI, may be another avenue to control cerebral edema and thereby mitigate intracranial hypertension and herniation.24 The success of these approaches will rest on their ability to prevent metabolic mismatch, avoid secondary injury, and improve long-term functional outcomes after severe TBI.
Ready to Claim Your Credits?
You have attempts to pass this post-test. Take your time and review carefully before submitting.
Good luck!
Recommended
- Alzheimer Disease & Dementias
Spirituality and Spiritual Care of People With Chronic Neurologic Illness
Sue Ouellette, PhD, MDivSue Ouellette, PhD, MDiv



