Maintaining Ambulation
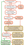
Multiple sclerosis (MS) is an autoimmune disease in which inflammatory processes damage myelin and axons in the central nervous system (CNS), producing lesions and leading to disrupted or disturbed neuronal signal transmission. Lesion activity, location, and load vary among individuals, as do the nature, severity, and variability of symptoms over time. A wide range of neurologic signs and symptoms associated with MS may, individually or cumulatively, result in worsening and potentially the eventual loss of ambulation. Motor weakness in the form of spasticity or motor fatigue, sensory dysfunction in proprioception or sensory touch, cognitive decline in ability to dual-task, visual problems including vision and eye movement control, and vestibular dysfunction caused by MS are among the many dysfunctions that can contribute to ambulation decline in in people with MS (Figure 1).
Frequency and Characteristics of Walking Limitations
Limitations in walking ability are common and well recognized in people with MS, and 75% of people with MS report walking impairment.1 Although limitations in walking are more frequent and severe later in the disease, they are also often present early.2,3 Changes in gait pattern, including reduced stride length, step width, stride time, swing-phase duration, and prolonged double-support time (Figure 2)4 as well as abnormal muscle-activation patterns and joint kinetics occur in people with MS compared with healthy persons.5 These changes can all interfere with walking speed, efficiency, and endurance. The ability to walk on uneven surfaces, slopes, in busy environments, or in the dark may also be affected by MS. There is an association between walking impairment and fall risk. Falls, along with their direct effects, may lead to lower levels of daily physical activity than recommended and curtailment of activities in general, both of which affect health, well-being, and participation in society.

Interventions to Maintain Walking
Disease-Modifying Therapies
Treatment of patients with MS comprises a combination of disease-modifying therapies (DMTs), relapse management, symptom management, and lifestyle wellness interventions. There are more than a dozen DMTs available, including injectable, oral, and infused medications that reduce the frequency of relapses in relapsing MS. Most of these also slow progression of disability in relapsing MS. Ocrelizumab also slows disability progression, including walking disability, in primary progressive MS. Appropriate use of DMTs is an essential component of maintaining ambulation for patients with MS. This article, however, focuses on additional approaches, including physical therapy, exercise, physical activity, and symptomatic medications, to further support maintaining ambulation in people with MS.
Physical Therapy
A variety of physical therapy interventions can improve several aspects of ambulation. Some focus on maintaining biomechanical prerequisites for ambulation, such as maintaining lower extremity joint range of motion to allow as normal a gait pattern as possible and thus prevent development of joint-related pain. Physical therapists also teach patients how to stretch muscles with increased tone to control spasticity and associated symptoms. Most commonly, however, physical therapy focuses on exercise to maintain and/or improve ambulation. Exercise as a component of physical therapy is delivered by a physical therapist who tailors and progresses therapy according to an individual’s abilities, limitations, goals, and resources. Exercise is supervised initially, with gradual progression to an independent home-exercise program. Home exercise is recommended in addition to supervised training to achieve more training during the period of physical therapy intervention and to prepare the patient for transition to ongoing independent exercise after the period of physical therapy.
A cluster of clinical trials has evaluated the effect of physical therapist-supervised exercise on ambulation in people with MS. These trials include similar elements, such as core stabilization, sensory strategies, dual tasking, treadmill or over-ground walking, general balance exercise, and flexibility exercises, either alone or in combination.6-10 The duration of interventions vary, from a 30-minute supervised exercise session once per week with 15 minutes daily home exercise for 12 weeks10 to 60-minute sessions twice per day for 5 consecutive days over 6 weeks that were supervised during the first 2 weeks.8 Despite differences in intervention content, quantity, and frequency, these studies support benefits of exercise delivered by a physical therapist on walking endurance, walking speed, and perceived walking limitations.
Aquatic therapy is also often provided by physical therapists and recommended for people with neurologic deficits. The body-weight support provided by water immersion can allow a person to perform movements that would otherwise be difficult or unsafe. Some evidence supports that aquatic therapy, provided as a 45- to 60-minute session 3 times per week, over a period of 8 weeks, can improve walking endurance in people with MS.11 The challenge of reaching and getting into and out of a pool can limit the application of aquatic therapy, however.
Interactive exergaming has also been evaluated as a physical therapy approach to promote exercise or training in people with MS but, in most of these trials, ambulation was not the primary outcome. A study in people with MS found that using either of 2 computerized stepping games for 30 minutes twice a week over a period of 12 weeks was associated with accelerated single- and dual-task gait speed as well as other positive outcomes.12 Other studied approaches to optimize outcomes from physical therapy include using virtual reality during treadmill training13 or using cooling garments during exercise.14 Exploring potentially complementary approaches may be the key to more effective interventions in the future.
A range of physical therapy interventions are associated with positive effects on ambulation in people with MS. The multifaceted nature of MS symptoms that may affect walking may, in part, explain why interventions targeting a combination of different aspects demonstrate success at the group level. However, when treating an individual, physical therapists may do better to tailor specific interventions to the specific abilities, limitations, and goals of that person. In addition, because MS is a lifelong and progressive disease, interventions will likely need to be modified over time to address changing needs and adherence.
Exercise
Exercise refers to a structured and repeated activity aiming to improve specific aspects of physical performance such as aerobic capacity, muscle strength, or balance.15 Resistance training has been shown to increase muscle strength16 as well as neural drive17 in people with MS; as muscle strength increases, walking speed increases.18 Exercise focused on improving balance can improve energy efficiency when walking, thereby reducing fatigue, while also helping reduce fear of falling and self-imposed walking restrictions. A systematic review with meta-analysis, which included only randomized controlled trials evaluating the effects of exercise on walking in people with MS, concluded that exercise was associated with increased walking speed and endurance.19 The greatest effects on walking speed were from interventions that combined resistance, aerobic, and balance exercise; the greatest effects on walking endurance were from combined training or resistance training alone.19 Most of these studies only included people with mild-to-moderate MS-associated disability. Although independent exercise without supervision can be beneficial, supervised exercise appears to be more effective than unsupervised exercise.20 Combining supervised and unsupervised exercise training periods may be ideal, promoting control and independence for the patient.
For people with MS, exercise should be individualized taking into consideration medical status and type, intensity, duration, and frequency of the intervention. In addition, professionals may also provide advice on how to manage spasticity, heat-sensitivity, or fatigue during exercise to optimize and enhance the benefits of an exercise program.
Physical Activity
Being physically active includes all bodily movement a person does, regardless of the goals, level of exertion, or duration of the activity.15 The Multiple Sclerosis Society of Canada has produced evidence-based physical activity guidelines for adults with MS. These guidelines recommend aerobic activity for 150 minutes per week at moderate intensity or 75 minutes per week at high intensity. In addition, they recommend twice weekly strength training activity for the major muscle groups that includes 1 to 2 sets of 10 to 15 repetitions of each exercise. Lower extremity activities are most likely to optimize benefits for ambulation.
Unfortunately, people with MS are less physically active than healthy people of the same age and sex and less active than is recommended by the guidelines.21 This inactivity contributes to physiologic deconditioning and may increase the impact of MS-related symptoms, including declines in, or even loss of, ambulation. Therefore, to help maintain their walking ability, in addition to exercise, we advise people with MS to be physically active and avoid a sedentary lifestyle.
Compensatory Strategies
In addition to physical therapy, other forms of exercise, physical activity, and compensatory strategies such as walking aids, orthoses, or technological devices like functional electrical stimulation (FES), can help maintain ambulation in people with MS. In FES, a transcutaneously delivered electrical current is used to stimulate peripheral motor nerves, eliciting muscle contractions, usually to produce ankle dorsiflexion during the swing phase of gait or to support hip and knee extension during stance phase (Figure 3). A systematic review of studies of FES in people with MS concluded that FES has positive effects on walking speed in people with MS during the time the device was applied. There was, however, no proven therapeutic carry-over effect when the device was not in use.22 The success of FES can depend on precise application of the electrodes, requiring some degree of skill and dexterity, and is not suitable for all people with MS. Some patients have or develop sensitivity to the skin adhesive on the electrodes, and extra caution is called for if there are sensory deficits in the area of stimulation. Rigid or nonrigid ankle-foot orthoses (AFOs) that support ankle dorsiflexion may be better tolerated by some individuals. Any of these strategies are likely to promote a smoother gait pattern reducing the energy needed to move the extremity forward. This may be especially important in patients with fatigue when walking longer distances or on uneven surfaces or if proprioception at the ankle is severely affected.

Although walking aids such as canes, crutches, trekking poles, or walkers can help to maintain ambulation, many people are reluctant to use a walking aid. This results in people performing below their ability to execute a task or action in a standard or uniform environment, or capacity. Recent research supports that walking-aid selection, fitting, and training by a physical therapist may help people with MS be more satisfied with their walking aid and promote them walking more, and more safely, with the walking aid.23
Symptomatic Medications
Various symptomatic medications can also help maintain walking in people with MS. Extended release dalfampridine is the only medication specifically approved by the Food and Drug Administration (FDA) to improve walking in patients MS. This approval was based on 2 phase 3 clinical trials demonstrating that approximately 35% to 40% of people with MS who took dalfampridine walked about 25% faster compared with patients with MS who took a placebo.24,25 Dalfampridine is potassium channel blocker administered as a 10 mg tablet to be taken twice a day approximately 12 hours apart. The effects of dalfampridine are symptomatic only with no known effect on MS progression or disease activity. Dalfampridine is contraindicated in people with hypersensitivity to dalfampridine, a history of seizure, or moderate or severe renal impairment (CrCl ≤ 50 mL/ min) and should be discontinued if a seizure occurs.
A number of other medications, although not specifically approved for this indication, may also improve walking in people with MS. For example, because walking in many people with MS is adversely affected by spasticity, antispasticity medications may improve walking. Baclofen is the antispasticity medication most commonly used in people with MS. It is generally well tolerated, and the dose can be titrated up as needed as underlying spasticity worsens. Tolerability of oral baclofen is generally limited by the dose-dependent side effect of fatigue. If this occurs, intrathecal baclofen pump implantation can be considered, although this comes with risk of complications that include pump failure or infection. Tizanidine, and occasionally benzodiazepines, are also used to help control MS-associated spasticity, although these can be associated with worse sedation than baclofen, and benzodiazepines come with the risks of tolerance and overdose. A number of studies have also demonstrated that cannabinoid preparations can reduce patient-rated spasticity.26 At this time none of these preparations are FDA approved in the US. Nabiximols (Sativex, GW Pharmaceuticals), is an oromucosal spray containing a 1:1 formulation of δ-tetrahydrocannibol (THC) and cannabidiol (CBD) that is approved for adjunctive treatment of MS-related spasticity outside the US.
In addition to antispasticity medications, medications that reduce pain or fatigue may also help maintain walking in some people with MS.
Involving the Patient
Onset of MS is typically in early adulthood, between the ages of 20 and 40 years with subsequent accumulation of disability over the person’s lifetime but with little impact on lifespan. It is, therefore, important to encourage people with MS to be physically active and exercise early in the disease course to optimize their long-term physical abilities. In our experience, with the right regimen from the start, ambulatory capacity may be maintained for longer. In Sweden, the Patient Act protects the rights and interests of patients and specifically states that each patient must be informed about their illness and the kinds of treatment available. They have the right to participate in all decisions about the care they will receive. Although this is not codified in law in the US, the ethical principle of autonomy requires health care providers to inform patients of their therapeutic options.27 Health care providers should strive for well-informed patients who know what they could potentially gain from exercising and being active. In addition, given the relatively short duration of direct contact with health care providers compared with the amount of time patients spend alone, we need robust methods to educate and facilitate patient engagement in their care and in follow-through with recommendations. Barriers to adherence with interventions to help maintain ambulation may include lack of confidence in recommended interventions, poor understanding or communication of specific recommendations, and limited access to recommended equipment and facilities. Telehealth and telerehabilitation may help support adherence from a distance, enabling those with access challenges to have ongoing support in following through with recommendations.28,29
Summary
Although decline of ambulatory capacity and performance are common and impactful in people with MS, a wide range of interventions have proven benefit or show promise for maintaining ambulation in this population. These include physical therapy, exercise, physical activity, and a range of both disease-modifying and symptomatic medications. Optimal outcomes depend on careful selection and modification of interventions by the health care team and use of effective strategies to involve the patient to optimize adherence.
1. Hobart JC, Riazi A, Lamping DL, Fitzpatrick R, Thompson AJ. Measuring the impact of MS on walking ability: the 12-Item MS Walking Scale (MSWS-12). Neurology. 2013;60(1):31-36.
2. Benedetti MG, Piperno R, Simoncini L, Bonato P, Tonini A, Giannini S. Gait abnormalities in minimally impaired multiple sclerosis patients. Mult Scler. 1999;5(5):363-368.
3. Martin CL, Phillips BA, Kilpatrick TJ, et al. Gait and balance impairment in early multiple sclerosis in the absence of clinical disability. Mult Scler. 2006;12(5):620-628.
4. Comber L, Galvin R, Coote S. Gait deficits in people with multiple sclerosis: systematic review and meta-analysis. Gait and Posture. 2017;51:25-35.
5. Cofre Lizama LE, Khan F, Lee PV, Galea MP. The use of laboratory gait analysis for understanding gait deterioration in people with multiple sclerosis. Mult Scler. 2016;22(14):1768-1776.
6. Cattaneo D, Jonsdottir J, Zocchi M, Regola A. Effects of balance exercises on people with multiple sclerosis: a pilot study. Clin Rehabilita. 2007;21(9):771-781.
7. Carling A, Forsberg A, Gunnarsson M, Nilsagard, Y. CoDuSe group exercise programme improves balance and reduces falls in people with multiple sclerosis: A multi-centre, randomized, controlled pilot study. Mult Scler. 2017;3(10):1394-1404.
8. Davies BL, Arpin DJ, Liu M, et al. Two different types of high-frequency physical therapy promote improvements in the balance and mobility of persons with multiple sclerosis. Arch Phys Med Rehabilita. 2016;97(12):2095-2101.e2093.
9. Forsberg A, von Koch L, Nilsagard, Y. Effects on balance and walking with the CoDuSe balance exercise program in people with multiple sclerosis: a multicenter randomized controlled trial. Mult Scler Int. 2016:7076265.
10. Kalron A, Rosenblum U, Frid L, Achiron A. Pilates exercise training vs. physical therapy for improving walking and balance in people with multiple sclerosis: a randomized controlled trial. Clin Rehabilita.2017;31(3):319-328.
11. Hoang P, Schoene D, Gandevia S, Smith S, Lord SR. Effects of a home-based step training programme on balance, stepping, cognition and functional performance in people with multiple sclerosis-a randomized controlled trial. Mult Scler. 2016;22(1):94-103.
12. Kargarfard M, Shariat, A, Ingle L, Cleland JA, Kargarfard M. Randomized controlled trial to examine the impact of aquatic exercise training on functional capacity, balance, and perceptions of fatigue in female patients with multiple sclerosis. Arch Phys Med Rehabilita. 2018:99(2):234-241.
13. Peruzzi A, Zarbo IR, Cereatti A, Della Croce U, Mirelman A. An innovative training program based on virtual reality and treadmill: effects on gait of persons with multiple sclerosis. Disabil Rehabilita. 2017;39(15):1557-1563.
14. Gonzales B, Chopard G, Charry B, Berger, E, Tripard J, Magnin E. Groslambert A. Effects of a Training Program Involving Body Cooling on Physical and Cognitive Capacities and Quality of Life in Multiple Sclerosis Patients: A Pilot Study. Europ Neurol. 2017;78(1-2):71-77.
15. Caspersen CJ, Powell KE, Christenson GM. Physical activity, exercise, and physical fitness: definitions and distinctions for health-related research. Pub Health Rep. 1985;100(2):126-131.
16. Dalgas U, Stenager E, Jakobsen J, et al. Resistance training improves muscle strength and functional capacity in multiple sclerosis. Neurology. 2009;73(18):1478-1484.
17. Dalgas U, Stenager E, Lund C, et al. Neural drive increases following resistance training in patients with multiple sclerosis. J Neurol. 2013;260(7):1822-1832.
18. Kjolhede T, Vissing K, Langeskov-Christensen D, et al. Relationship between muscle strength parameters and functional capacity in persons with mild to moderate degree multiple sclerosis. Mult Scler Relat Disord. 2015;4(2):151-158.
19. Pearson M, Dieberg G, Smart N. Exercise as a therapy for improvement of walking ability in adults with multiple sclerosis: a meta-analysis. Arch Phys Med Rehabilita. 2015;96(7):1339-1348.e1337.
20. Dalgas U, Stenager E, Ingemann-Hansen T. Multiple sclerosis and physical exercise: recommendations for the application of resistance-, endurance- and combined training. Mult Scler. 2008;14(1):35-53.
21. Kinnett-Hopkins D, Adamson B, Rougeau K., Motl RW. People with MS are less physically active than healthy controls but as active as those with other chronic diseases: An updated meta-analysis. Mult Scler Relat Disord. 2017;13:38-43.
22. Springer S, Khamis S. Effects of functional electrical stimulation on gait in people with multiple sclerosis - A systematic review. Mult Scler Relat Disord. 2017;3:4-12.
23. Martini DN, Zeeboer E, Hildebrand A, et al. ADSTEP: Preliminary investigation of a multicomponent walking aid program in people With multiple sclerosis. Arch Phys Med Rehabilita. 2018;9(10):2050-2058.
24. Goodman AD, Brown TR, Edwards KR, et al. A phase 3 trial of extended release oral dalfampridine in multiple sclerosis. Ann Neurology, 2010;68(4):494-502.
25. Goodman AD, Brown TR, Krupp LB, et al. Sustained-release oral fampridine in multiple sclerosis: a randomised, double-blind, controlled trial. Lancet. 2009;373(9665):732-738.
26. Torres-Moreno, M., Papaseit E, Torrens M, Farré M. Assessment of efficacy and tolerability of medicinal cannabinoids in patients with multiple sclerosis: A systematic review and meta-analysis. JAMA Network Open. 2018;1(6):e183485.
27. Beauchamp TL, Childress JF. Principles of Biomedical Ethics. 7th ed. Oxford, England, UK: Oxford University Press; 2012.
28. Giunti G, Kool J, Rivera Romero O, Dorronzoro Zubiete, E. Exploring the specific needs of persons with multiple sclerosis for health solutions for physical activity: mixed-methods Study. JMIR Mhealth Uhealth. 2018;6(2):e37.
29. Thirumalai M, Rimmer JH, Johnson G, et al. TEAMS (Tele-Exercise and Multiple Sclerosis), a tailored telerehabilitation mHealth app: participant-centered development and usability study. JMIR Mhealth Uhealth. 2018;6(5):e10181.
Disclosures
MHC has received consulting fees from GW Pharmaceuticals. YEN has nothing to disclose.
Ready to Claim Your Credits?
You have attempts to pass this post-test. Take your time and review carefully before submitting.
Good luck!
Recommended
- MS & Immune Disorders
Ocrevus Treatment in Pregnant and Lactating Women with Multiple Sclerosis
Riley Bove, MD, MMScRiley Bove, MD, MMSc - Alzheimer Disease & Dementias
Capacity Determination and Advance Care Planning
Michael Rubin, MD, MA; Anne Lai Howard, JDMichael Rubin, MD, MA; Anne Lai Howard, JD



